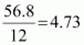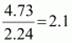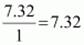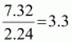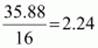Basic Concepts of Chemistry
Unit System
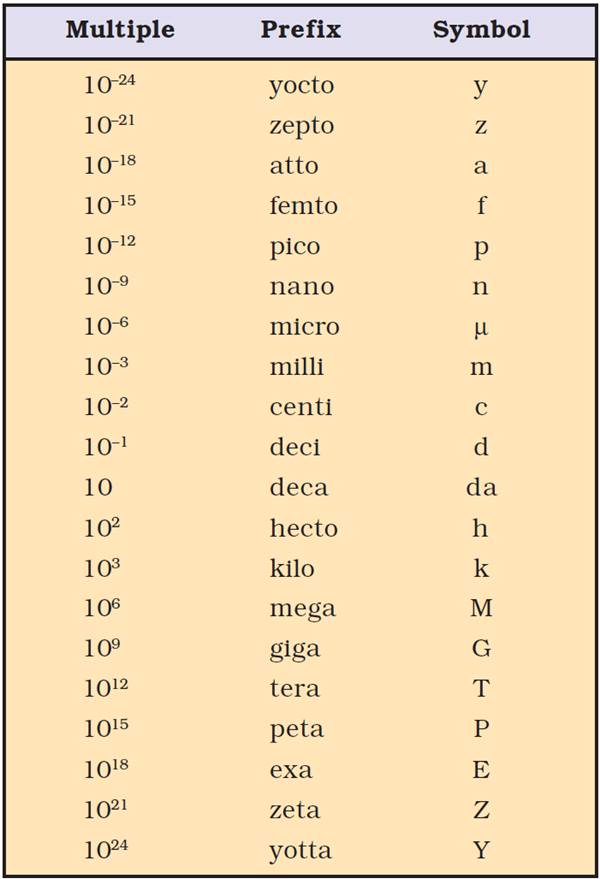
Common prefixes used in SI system:
Accuracy and Precision
Accuracy denotes the closeness of an experimental value or the mean value of a set of measurements to the true value. Thus,
Accuracy is a measure of the difference between the experimental value or the mean value of a set of measurements and the true value.
Precision refers to how closely two or more measurements of the same quantity agree with one another. Thus,
Precision is expressed as the difference between a measured value and the arithmetic mean value for a series of measurements.
Example:
Results obtained by four students for the percentage of iron in a sample.
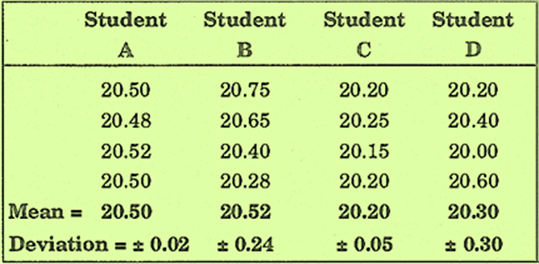
The results obtained by student A have good precision because the individual measurements are close together (± 0.02) and also have good accuracy because the mean result is same as the true value.
The results of student B have low or poor precision because the
measurements are scattered
(± 0.24) but have good accuracy because the mean value (20.52) is close to the
true value.
Student C has obtained results of good precision because the measurements are close together (± 0.05) but have poor accuracy because the mean value (20.20) is far from the true value.
The results of student D have poor precision because the measurements are scattered and have poor accuracy because the mean value (20.30) is far from the true value.
Note: Good precision does not assure good accuracy.
Significant Figures
The significant figures in a number are all the certain digits plus one uncertain digit.
For example, in the above value (9.265 g), there are four significant figures.
Thus, the number of significant figures conveys the information that except the last digit, all other digits are known with certainty.
Rules for Determining the Number of Significant Figures
(i) All non-zero digits are significant. For example, 17.182 has five significant figures
(ii) Zeros between two non-zero digits are significant. For example, 5.02 has three significant figures
(iii) The zeros preceding to the first non-zero number (i.e., to the left of the first non-zero number) are not significant. Such zeros indicate the position of decimal point. For example, 0.125 has three significant figures, and 0.0071 has two significant figures
(iv) All zeros placed at the end or to the right of a number are significant provided they are on the right side of the decimal point. In fact, these represent the accuracy or the precision of the measuring scale. For example, 0.0600 has three significant figures, and 354.00 has five significant figures
(v) The results obtained by counting are exact numbers i. e., numbers· without any uncertainty. These exact numbers have infinite number of significant figures. For example, 2 balls or 20 eggs, there are infinite number significant figures. because these are exact numbers-and may be represented by writing infinite number of zeros after placing a decimal i.e., 2 = 2.000000 or 20 = 20.000000.
(vi) Zeros at the end of a number without decimal point are not significant.
Temperature
There are three common scales to measure temperature — °C (degree Celsius), °F (degree Fahrenheit) and K (kelvin). Here, K is the SI unit.
The temperatures on two scales are related to each other by the following relationship:
°F =  (°C) + 32
(°C) + 32
The kelvin scale is related to Celsius scale as follows:
K = ℃ + 273.15
Example: The boiling point of water at sea level is 212 °F. What is its equivalent in Kelvin scale?
Solution: To convert temperature from Fahrenheit scale into Kelvin scale, the following equations are used.
°F =  (°C) + 32
(°C) + 32
°C = (°F − 32)/1.8
K = °C + 273.15
Converting °F into °C,
°C = (212 − 32)/1.8
= 100
Therefore, the boiling point of water is 100°C converting °C into K.
K = °C + 273.15
= 100 + 273.15 = 373.15
Hence, 212°F is equivalent to 373.15 K.
Law of Chemical Combinations
The combination of elements to form compounds is governed by the following five basic laws.
Law of Conservation of Mass
Matter can neither be created nor destroyed.
This was the result of exact measurement of masses of reactants and products.
Law of Definite Proportions
A given compound always contains exactly the same proportion of elements by weight.
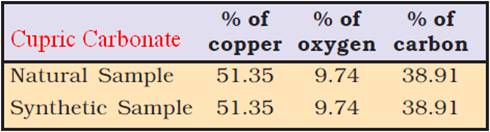
Law of Multiple Proportions
If two elements can combine to form more than one compound, the masses of one element that combine with a fixed mass of the other element, are in the ratio of small whole numbers.
Put forth by John Dalton (1803).
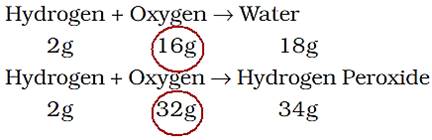
Law of Gaseous Volumes
When gases combine or are produced in a chemical reaction, they do so in a simple ratio by volume provided all gases are at same temperature and pressure.
Put forth by Gay Lussac (1808).
Thus, 100 mL of hydrogen combine with 50 mL of oxygen to give 100 mL of water vapour.
Hydrogen + Oxygen → Water
100 mL 50 mL 100 mL
Thus, the volumes of hydrogen and oxygen which combine together (i.e., 100 mL and 50 mL) bear a simple ratio of 2:1.
Gay-Lussac’s discovery of integer ratio in volume relationship is actually the law of definite proportions by volume. The law of definite proportions, stated earlier, was with respect to mass. The Gay-Lussac’s law was explained properly by the work of Avogadro in 1811.
Avogadro Law
Equal volumes of gases at the same temperature and pressure should contain equal number of molecules.
Put forth by Avogadro (1811).
Avogadro made a distinction between atoms and molecules which is quite understandable in the present times. If we consider again the reaction of hydrogen and oxygen to produce water, we see that two volumes of hydrogen combine with one volume of oxygen to give two volumes of water without leaving any unreacted oxygen.
Application of Avogadro’s Law
Application of Avogadro Law can deduce that
(i) Molecular Mass = 2 x Vapour Density
Where,
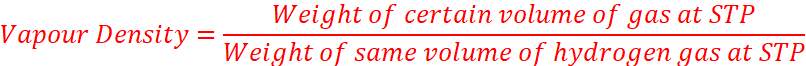
(ii) 22.4 L of any gas at STP weigh equal to Molecular Mass expressed in grams. This mass is known as Gram Molecular Mass, and this volume is called Gram Molecular Volume (GMV).
STP: 0°C and 1 atm.
NB: If conditions taken are 1 bar and 0°C then it will be 22.7 L.
Dalton’s Atomic Theory (1808)
1. Matter consists of indivisible atoms.
2. All the atoms of a given element have identical properties including identical mass. Atoms of different elements differ in mass.
3. Compounds are formed when atoms of different elements combine in a fixed ratio.
4. Chemical reactions involve reorganization of atoms. Atoms are neither created nor destroyed in a chemical reaction.
Dalton’s theory was able to explain the laws of chemical combination (except Avogadro law).
Atomic Mass
Atomic Mass is the mass of an atom of a chemical element expressed in atomic mass units.
Atomic Mass Unit (amu, now replaced by unified mass, u)
It is equal to 1/12th of the mass of an atom of 12C isotope, and is equal to 1.66054 × 10-24 g.
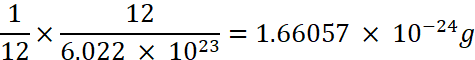
Average Atomic Mass
It is the average of atomic masses of various isotopes of an element as per its abundance.
Average atomic mass = ∑(Mass of isotope × Relative abundance)
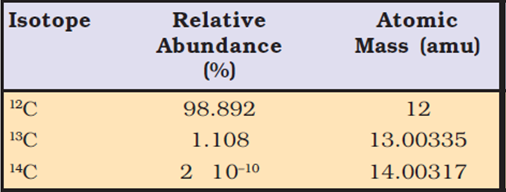
From the above data, the average atomic mass of carbon will come out to be:
(0.98892) (12 u) + (0.01108) (13.00335 u) + (2 × 10–12) (14.00317 u)
= 12.011 u
Example
The relative abundance of two isotopes of copper, having atomic masses 62.93 u and 64.94 u, are 69.09% and 30.91% respectively. Calculate the average atomic mass of copper.
Solution:
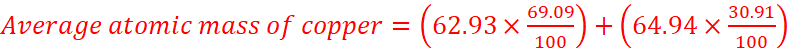
= 63.55 u
Example: Calculate the average atomic weight of silicon having 92.2% of Si-28 isotope of relative mass 27.98 amu, 4.7% of Si-29 isotope of relative mass 28.98 amu and 3.1% of Si-30 isotope of relative mass 29.97 amu.
Solution: Given: 92.20% of silicon of mass 27.98 amu
4.7% of silicon of mass 28.98 amu
3.1 % of silicon of mass 29.97 amu
Average atomic weight =
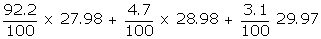
= 25.80 + 1.36 + 0.93
= 28.09 amu.
Example: Average atomic weight of silicon is 28.09 amu. One million silver atoms weigh 1.79 x 1016 g. Calculate the gram atomic mass of silver.
Solution: Number of silver atoms = 1 million = 1 × 106
Mass of one million Ag atoms = 1.79 × 1016 g
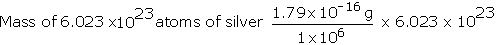
= 107.8 g
Gram atomic mass of silver is equal to the mass of 6.023 × 1023 atoms of silver. So, the gram atomic mass of silver is 107.8 g
Example: Chlorophyll, the green colouring matter of plants responsible for photosynthesis, contains 2.68% of magnesium by weight. Calculate the number of Mg atoms in 2.00 g of chlorophyll.
Solution: 100 g of chlorophyll contains 2.68 g of Mg
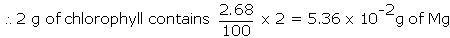
1 mole of Mg = 24 g of Mg = 6.023 x 1023 atom of Mg
24 g of Mg contains, 6.023 × 1023 atom of Mg
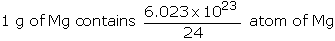
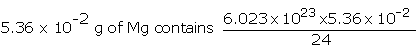
=1.34 × 1021 Mg atoms
Example: A sample of gaseous substance weighing 0.5 g occupies a volume of 1.12 litres under STP conditions. Calculate the molar mass of the substance.
Solution: 1 mole of any gaseous substance at STP occupies 22.4 L.
1.12 L of gaseous substance = 0.5 g
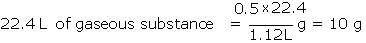
The molar mass of the substance therefore is 10 g/mol.
Molecular Mass
Molecular mass is the sum of atomic masses of the elements present in a molecule.
Example: Molecular mass of methane, (CH4)
= (12.011 u) + 4 (1.008 u)
= 16.043 u
Similarly, molecular mass of water (H2O)
= 2 × atomic mass of hydrogen + 1 × atomic mass of oxygen
= 2 (1.008 u) + 16.00 u
= 18.02 u
Example: Molecular mass of CO2 = 1 × Atomic mass of carbon + 2 × Atomic mass of oxygen
= (1 × 12.011 u) + (2 × 16.00 u)
= 12.011 u + 32.00 u
= 44.011 u
Formula Mass
For the substances which do not contain molecules, rather they possess +ve and –ve ions, the mass is calculated based on their formula, e.g., NaCl. The method is same as that for Molecular Mass.
Example: Formula mass of sodium chloride (NaCl)
= Atomic mass of sodium + Atomic mass of chlorine
= 23.0 u + 35.5 u
= 58.5 u
Example: 0.158 g of an acid acquired 40 mL of 0. 2 N KOH for complete neutralization. Calculate the equivalent mass of the acid.
Let the equivalent mass of the acid be E.
Gram equivalent of acid = 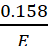
Gram equivalent of acid = Gram equivalent of base
Or 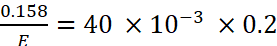
where, E is the equivalent mass of the acid.
Hence, E = 19.75 g
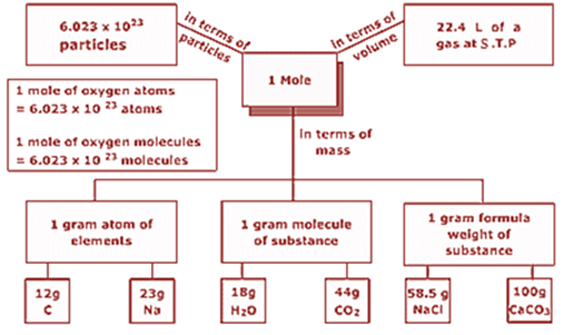
Mole Concept and Molar Masses
Mole
1 mole of any substance can be defined as:
Amount of a substance that contains as many particles (atoms, molecules or ions) as there are atoms in 12 g of the 12C isotope
Avogadro number or Avogadro constant (NA); equal to 6.022 × 1023 particles
Example:
1 mole of hydrogen atoms = 6.022 × 1023 atoms
1 mole of carbon dioxide molecules = 6.022 × 1023 molecules
1 mole of sodium chloride = 6.022 × 1023 formula units of sodium chloride
Molar mass of a substance can be defined as:
Mass of one mole of a substance in grams.
It is numerically equal to atomic/molecular/formula mass in u.
Example
Molar mass of CO2 = 44.011 g mol−1
Molar mass of NaCl = 58.5 g mol−1
Example What number of moles contains 3.011 × 1023 molecules of glucose?
Solution:
1 mole of glucose is equivalent to 6.022 × 1023 molecules of glucose.
Hence, 3.011 × 1023 molecules of glucose will be present in
= 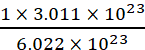 mol = 0.5 mol (of glucose)
mol = 0.5 mol (of glucose)
Thus, 0.5 mole of glucose contains 3.011 × 1023 molecules of glucose.
Example What is the mass of a mole of fluorine molecule?
Solution: 1 mole of fluorine molecule contains 6.022 × 1023 molecules and weighs 38 g.
Therefore, mass of a fluorine molecule = 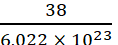 g
g
= 6.31 × 10−23 g
Percentage Composition
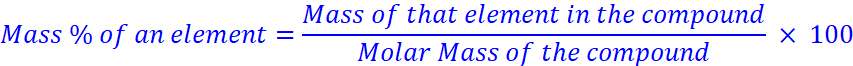
Example What is the mass percent of oxygen in potassium nitrate? (Atomic mass of K = 39.10 u, atomic mass of N = 14.007 u, atomic mass of O = 16.00 u)
Solution: Atomic mass of K = 39.10 u (Given)
Atomic mass of N = 14.007 u (Given)
Atomic mass of O = 16.00 u (Given)
Therefore, molar mass of potassium nitrate (KNO3)
= 39.10 + 14.007 + 3(16.00)
= 101.107 g
Therefore, mass percent of oxygen in KNO3
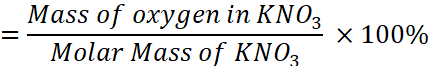
= 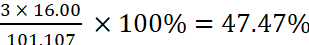
Empirical Formula and Molecular Formula
|
Empirical formula |
Molecular formula |
|
Represents the simplest whole number ratio of various atoms present in a compound |
Represents the exact number of different types of atoms present in a molecule of a compound |
Empirical formula is determined if mass % of various elements are known.
Molecular formula is determined from empirical formula if molar mass is known.
Example: A compound contains 92.26% carbon and 7.74% hydrogen. If the molar mass of the compound is 26.038 g mol−1, then what are its empirical and molecular formulae?
Solution: Mass percent of carbon (C) = 92.26% (Given)
Mass percent of hydrogen (H) = 7.74% (Given)
Number of moles of carbon present in the
compound = 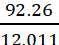 = 7.68 mol
= 7.68 mol
Number of moles of hydrogen present in the
compound = 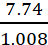 = 7.68 mol
= 7.68 mol
Thus, in the given compound, carbon and hydrogen are present in the ratio C : H = 7.68 : 7.68
= 1 : 1
Therefore, the empirical formula of the compound is CH.
Empirical formula mass of CH = (12.011 + 1.008) g = 13.019 g
Molar mass of the compound = 26.038 g (Given)
Therefore, n = 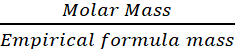 =
= 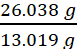 = 2
= 2
Hence, the molecular mass of the compound is (CH)n, i.e., (CH)2 or C2H2.
Example: An organic compound contains 56.8 % carbon, 7.32 % hydrogen and the remaining oxygen. The vapour density of the compound is 68. Calculate the molecular formula of the compound.
|
Element |
Percentage |
Atomic mass |
Moles of atom |
Atomic ratio |
Simplest ratio |
|
C |
56.8 |
12 |
|
|
2 |
|
H |
7.32 |
1 |
|
|
3 |
|
O |
35.88 |
16 |
|
|
1 |
∴ Empirical formula = C2H3O
Empirical formula mass = 12 × 2 + 1 × 3 + 16 × 1= 43 g
Molecular mass = 2 × Vapour density
= 2 × 68 = 136
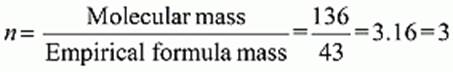
Molecular formula = (E.F.)n = (C2H3O)3 = C6H9O3
Interconversion among number of moles, mass and number of molecules
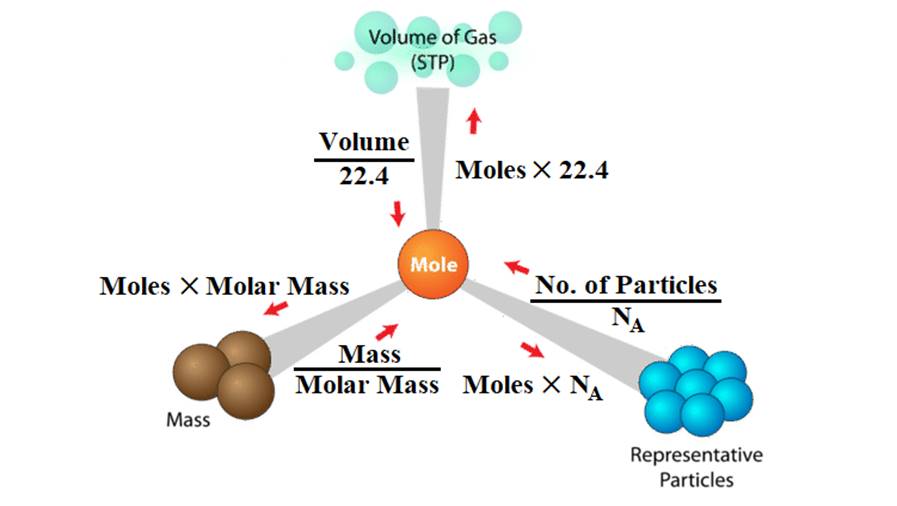
Example: Calculate the number of moles of HCl left if 2.98 × 1020 molecules are removed from 36.5 mg of HCl.
Solution:
Moles of HCl removed = 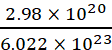 = 0.49 × 10−3
= 0.49 × 10−3
Total moles of HCl = 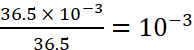
Moles of HCl left = 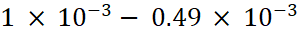
= 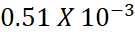
Example: Calculate the least molecular weight of DNA if it contains 4.5 % nitrogen.
Solution: For the least molecular mass of DNA, it must have at least one nitrogen atom in its one molecule.
If it has 4.5 g N, the mol mass = 100 g
If it has 14 g N, the mol mass = 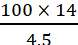 g = 311.11 g
g = 311.11 g
Example: The vapour density of a mixture containing NO2 and N2O4 is 38.3 at 27° C. Calculate the number of moles of NO2 in 100 g of the mixture.
Solution: Molecular mass of mixture = 2 × Vapour density = 2 × 38.3 = 76.6
Let us assume that x g of NO2 is present in 100 g mixture
∴ N2O4 = (100 − x) g
Total number of moles of mixture = No. of moles of NO2 + No. of mole of N2O4
Or 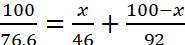
Or x = 20.10 g
Moles of NO2 in mixture 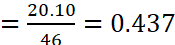
Stoichiometry and Stoichiometric Calculations
Stoichiometry deals with the calculation of masses (sometimes volumes also) of the reactants and the products involved in a chemical reaction.
- An example of a balanced chemical equation is given below.

From the above balanced chemical equation, the following information is obtained:
- One mole of C3H8(g) reacts with five moles of O2(g) to give three moles of CO2(g) and four moles of H2O(l).
- One molecule of C3H8(g) reacts with five molecules of O2(g) to give three molecules of CO2(g) and four molecules of H2O(l).
- 44 g of C3H8(g) reacts with (5 × 32 = 160) g of O2(g) to give (3 × 44 = 132) g of CO2 and (4 × 18 = 72) g of H2O.
- 22.4 L of C3H8(g) reacts with (5 × 22.4) L of O2(g) to give (3 × 22.4) L of O2 and (4 × 22.4) L of H2O.
Example: Nitric acid (HNO3) is commercially manufactured by reacting nitrogen dioxide (NO2) with water (H2O). The balanced chemical equation is represented as follows:
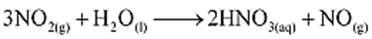
Calculate the mass of NO2 required for producing 5 moles of HNO3.
Solution: According to the given balanced chemical equation, 3 moles of NO2 will produce 2 moles of HNO3.
Therefore, 2 moles of HNO3 require 3 moles of NO2.
Hence, 5 moles of HNO3
require = 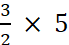 moles of NO2 = 7.5 moles of NO2
moles of NO2 = 7.5 moles of NO2
Molar mass of NO2 = (14 + 2 × 16) g mol−1 = 46 g mol−1
Thus, required mass of NO2 = (7.5 × 46) g mol−1 = 345 g mol−1
Limiting Reagent
- Reactant which gets completely consumed when a reaction goes to completion
- Limits the amount of the product formed
Example: Lead nitrate reacts with sodium iodide to give lead iodide and sodium nitrate in the following manner:

What amount of sodium nitrate is obtained when 30 g of lead nitrate reacts with 30g of sodium iodide?
Solution:
Molar mass of
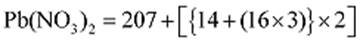
= 331 g mol−1
Molar mass of NaI = (23 + 127) = 150 g mol−1
According to the given equation, 1 mole of Pb(NO3)2 reacts with 2 moles of NaI, i.e.,
331 g of Pb(NO3)2 reacts with 300 g of NaI to give PbI2 and NaNO3
Thus, Pb(NO3)2 is the limiting reagent.
Therefore, 30 g of Pb (NO3)2
= 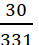 = 0.09 mole
= 0.09 mole
According to the equation, 0.09 mole of Pb(NO3)2 will give (2 × 0.09) mole of NaNO3 = 0.18 mole of NaNO3.
Mass Percent (also termed as Weight Percent) w/w%

Example
4.4 g of oxalic acid is dissolved in 200 mL of a solution. What is the mass per cent of oxalic acid in the solution? (Density of the solution = 1.1 g mL−1)
Solution:
Density of the solution = 1.1 g mL−1
So, the mass of the solution = (200 mL) × (1.1 g mL−1)
= 220 g
Mass of oxalic acid = 4.4 g
Therefore, mass per cent of oxalic acid in the solution

Mole Fraction
If a substance ‘A’ dissolves in a substance ‘B’, then mole fraction of A
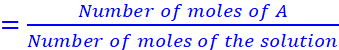
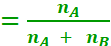
Mole fraction of B
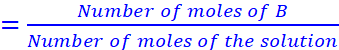
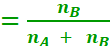
nA − Number of moles of A
nB − Number of moles of B
Example: A solution is prepared by dissolving 45 g of a substance X (molar mass = 25 g mol−1) in 235 g of a substance Y (molar mass = 18 g mol−1). Calculate the mole fractions of X and Y.
Solution:
Moles of X, nX = 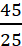
= 1.8 mol
Moles of Y, nY = 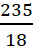
= 13.06 mol
Therefore, mole fraction of X, nX
= 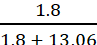 = 0.121
= 0.121
And, mole fraction of Y, nY = 1 − nX
= 1 − 0.121 = 0.879
Molarity
Number of moles of a solute in 1 L of a solution

Molarity equation:
M1V1 = M2V2
M1 = Molarity of a solution when its volume is V1
M2 = Molarity of the same solution when its volume is V2
Examples: 10 g of HCl is dissolved in enough water to form 500 mL of the solution. Calculate the molarity of the solution.
Solution: Molar mass of HCl = 36.5 g mol−1
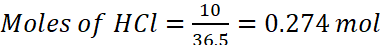
Volume of the solution = 500 mL = 0.5 L
Therefore, molarity =

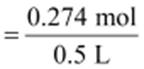
= 0.548 M
Example: Commercially available concentrated HCl contains 38% HCl by mass. What volume of concentrated HCl is required to make 2.5 L of 0.2 M HCl? (Density of the solution = 1.19 g mL−1)
Solution: 38% HCl by mass means that 38g of HCl is present in 100 g of the solution.
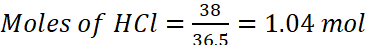
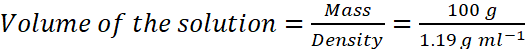
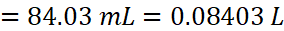
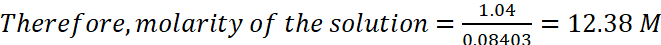
According to molarity equation,
M1V1 = M2V2
Here,
M1 = 12.38 M, M2 = 0.2 M, V2 = 2.5 L
Now, M1V1 = M2V2
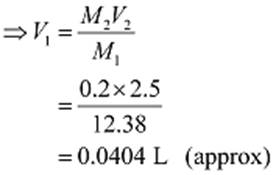
Hence, required volume of HCl = 0.0404 L
Molality
Number of moles of solute present in 1 kg of solvent
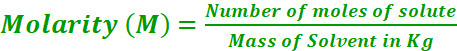
Example: What is the molality of a solution of glucose in water, which is labelled as 15% (w/w)?
Solution: 15% (w/w) solution means that 15 g of glucose is present in 100 g of the solution, i.e., (100 − 15) g = 85 g of water = 0.085 kg of water
Moles of glucose = 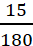 = 0.083 mol
= 0.083 mol
Therefore, molality of the solution = 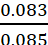 = 0.976 m
= 0.976 m
Online Tuitions & Self−Study Courses for Grade 6 to 12 & JEE / NEET