JEE NEET Chemistry
Chemical Bonding
Important Questions
Q.1. The state of hybridization of the central atom and the number of lone pairs over the central atom in POCl3 are
(a) sp, 0 (b) sp2, 0 (c) sp3, 0 (d) dsp2, 1
Q.2. The paramagnetic behaviour of B2 is due to the presence of
(a) 2 unpaired electrons in πb MO
(b) 2 unpaired electrons in π* MO
(c) 2 unpaired electrons in σ* MO
(d) 2 unpaired electrons in πb MO
Q.3. Which one of the following contains ionic, covalent and co−ordinate bonds?
(a) NaOH
(b) NaCl
(c) NaCN
(d) NaNC
Q.4. In which of the following pairs the two species are not isostructural?
(a) PCl4+ and SiCl4
(b) PF5 and BrF5
(c) AlF63− and SF6
(d) CO32− and NO3−
Q.5. The increasing order of O−N−O bond angle in the species NO2, NO2+ and NO2− is
(a) NO2+ < NO2 < NO2−
(b) NO2− < NO2 < NO2+
(c) NO2+ < NO2− < NO2
(d) NO2 < NO2+ < NO2−
Q.6. Molecular shapes of SF4, CF4 and XeF4 and the number of lone pairs on the central atom are respectively.
(a) the same, with 1, 2 and 1
(b) the same, with 1, 0 and 1
(c) different, with 0, 1 and 2
(d) different, with 1, 0 and 2
Q.7. Allyl cyanide molecule contains
(a) 9 sigma bonds, 4 pi bonds and no lone pair
(b) 9 sigma bonds, 3 pi bonds and one lone pair
(c) 8 sigma bonds, 5 pi bonds and one lone pair
(d) 8 sigma bonds, 3 pi bonds and two lone pairs
Q.8. In which of the following pairs of molecules/ions, both the species are not likely to exist?
(a) H2−, He22+
(b) H2+, He22−
(c) H2−, He22−
(d) H22+, He2
Q.9. Stability of the species Li2, Li2− and Li2+ increases in the order of
(a) Li2− < Li2 < Li2+
(b) Li2 < Li2+ < Li2−
(c) Li2− < Li2+ < Li2
(d) Li2 < Li2− < Li2+
Q.10. The structure of XeF6 is experimentally determined to be distorted octahedron. Its structure according to VSEPR theory is
(a) octahedron
(b) trigonal bipyramid
(c) pentagonal bipyramid
(d) tetragonal bipyramid
Q.11. The bond angle in NF3 (102.3°) is smaller than NH3 (107.2°). This is because of
(a) large size of F compared to H
(b) large size of N compared to F
(c) opposite polarity of N in the two molecules
(d) small size of H compared to N
Q.12. The lattice energy of NaCl is 788 kJ mol−1. This means that 788 kJ of energy is required
(a) to separate one mole of solid NaCl into one mole of Na+(g) and one mole of Cl−(g) to infinite distance
(b) to separate one mole of solid NaCl into one mole of Na(g) and one mole of Cl(g) to infinite distance
(c) to convert one mole of solid NaCl into one mole of gaseous NaCl
(d) to convert one mole of gaseous NaCl into one mole of solid NaCl
Q.13. Molecular formulae and shapes of some molecules are given below. Choose the incorrect match.
|
Formula |
Shape |
|
(a) NH3 |
Trigonal pyramidal |
|
(b) SF4 |
Tetrahedral |
|
(c) ClF3 |
T−shaped |
|
(d) PCl5 |
Trigonal bipyramidal |
|
(e) BF3 |
Trigonal planar |
Q.14. The pair of compounds having identical shapes for their molecules is
(a) CH4, SF4
(b) BCl3, ClF3
(c) XeF2, ZnCl2
(d) SO2, CO2
Q.15. The correct arrangement of species in the decreasing order of the bond length between carbon and oxygen in them is
(a) CO, CO2, HCO2−, CO32−
(b) CO2, HCO2−, CO, CO32−
(c) CO32−, HCO2−, CO2, CO
(d) CO, CO32−, CO2, HCO2−
Q.16. Which one of the following properties is not shown by NO?
(a) Its bond order is 2.5
(b) It is diamagnetic in gaseous state
(c) It is a neutral oxide
(d) It combines with oxygen to form nitrogen dioxide
Q.17. The number of lone pairs of electrons on the central atoms of H2O, SnCl2, PCl3 and XeF2 respectively, are
(a) 2, 1, 1, 3
(b) 2, 2, 1, 3
(c) 3, 1, 1, 2
(d) 2, 1, 2, 3
Q.18. The correct order of O−O bond length in O2, H2O2 and O3 is
(a) O2 > O3 > H2O2
(b) H2O2 > O3 > O2
(c) O3 > O2 > H2O2
(d) O3 > H2O2 > O2
Q.19. Identify the T−shaped molecule in the following:
(a) BF3
(b) NH3
(c) NF3
(d) ClF3
Q.20. Which one of the following molecule has the least dipole moment?
(a) H2O
(b) BeF2
(c) NH3
(d) NF3
Q.21. Main axis of diatomic molecule is z −axis. The orbitals Px and Py overlap to form
(a) π−molecular orbital
(b) δ−molecular orbital
(c) δ−molecular orbital
(d) no bond is formed
Q.22. The increasing order of bond orders of O2, O2+, O2− and O22− is
(a) O2+, O2, O2−, O22−
(b) O22−, O2−, O2+, O2
(c) O2, O2+, O2−, O22−
(d) O22−, O2−, O2, O2+
Q.23. The species in which the N atom in a state of sp hybridisation is
(a) NO2+
(b) NO2−
(c) NO3−
(d) NO2
Q.24. The ground state magnetic property of B2 and C2 molecules will be
(a) B2 paramagnetic and C2 diamagnetic
(b) B2 diamagnetic and C2 paramagnetic
(c) both are diamagnetic
(d) both are paramagnetic
Q.25. Which of the following structures of a molecule is expected to have three bond pairs and one lone pair of electrons?
(a) Trigonal Planar
(b) Tetrahedral
(c) Octahedral
(d) Pyramidal
Q.26. Which of the following is the correct electron dot structure of N2O molecule
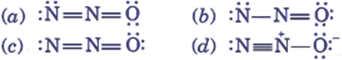
Q.27. Among the following, the molecule that will have the highest dipole moment is
(a) H2
(b) HI
(c) HBr
(d) HF
Q.28. Which of the following pairs have identical bond order?
(a) CN− and NO+
(b) CN− and O2−
(c) CN− and CN+
(d) NO+ and O2−
Q.29. Which of the following species is not paramagnetic?
(a) NO
(b) CO
(c) O2
(d) B2
Q.30. What will be the geometry of the compound MB4L2? Here B is bond pair and L is lone pair.
(a) Square planar
(b) Octahedral
(c) Square pyramid
(d) Tetrahedral
Q.31. What is the hybridisation and geometry of the given species? The species are XeF2 and ICl2−.
(a) sp3d and trigonal bipyramidal
(b) sp3d2 and square planar
(c) sp3d and linear
(d) sp3 and irregular tetrahedron
Q.32. Which of the following has the strongest H−bond?
(a) O−H−S
(b) S−H−O
(c) F−H−F
(d) F−H−O
Q.33. The intramolecular hydrogen bond is present in
(a) phenol
(b) o−nitrophenol
(c) p−nitrophenol
(d) p−cresol
Q.34. The state of hybrid orbitals of carbon in CO2, CH4 and CO32− respectively is
(a) sp3, sp2 and sp
(b) sp3, sp and sp2
(c) sp, sp3 and sp2
(d) sp2, sp3 and sp
Q.35. According to molecular orbital theory, which of the following is not a viable molecule ?
(a) He22+
(b) He2+
(c) H2−
(d) H22−
Q.36. Which of the following compounds contain(s) no covalent bond(s)?
KCl, PH3, O2, B2H6, H2SO4
(a) KCl, B2H6, PH3
(b) KCl, H2SO4
(c) KCl
(d) KCl, B2H6
Q.37. Total number of lone pair of electrons in I3− ion is:
(a) 3
(b) 6
(c) 9
(d) 12
Q.38. According to molecular orbital theory, which of the following is true.?
(a) Both Li2+ and Li2− are unstable
(b) Li2+ is unstable and Li2− is stable
(c) Li2+ is stable and Li2− is unstable
(d) Both Li2+ and Li2− are stable
Q.39. In which of the following processes, the bond order has increased and paramagnetic character has changed to diamagnetic ?
(a) N2 → N2+
(b) NO → NO+
(c) O2 → O22+
(d) O2 → O2+
Q.40. Total pi and half sigma bonds are present in
(a) N2+
(b) N2
(c) O2+
(d) O2
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
