Q.1. Rate constant k for a first order reaction has been found to be 2.54 × 10−3 sec −1. Calculate its 3/4th life. (log 4 = 0.6020).
Q.2. For the first order thermal decomposition reaction, the following data were obtained:
C2H5Cl(g) → C2H4(g) + HCl(g)
|
Time/sec |
Total pressure/atm |
|
0 |
0.30 |
|
300 |
0.50 |
Calculate the rate constant.
(Given : log 2 = 0.301, log 3 = 0.4771, log 4 = 0.6021)
Q.3. A first order reaction takes 30 minutes for 50% completion. Calculate the time required for 90% completion of this reaction. (log 2 = 0.3010)
Q.4. A first order reaction takes 10 minutes for 25% decomposition. Calculate t1/2 for the reaction. (Given : log 2 = 0.3010, log 3 = 0.4771, log 4 = 0.6021)
Q.5. For a chemical reaction R → P, the variation in the concentration, ln [R] vs. time (s) plot is given as
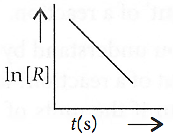
(i) Predict the order of the reaction.
(ii) What is the slope of the curve?
(iii) Write the unit of the rate constant for this reaction.
Q.6. The rate constant for a first order reaction is 60 s−1. How much time will it take to reduce the initial concentration of the reactant to its 1/10th value?
Q.7. The following data were obtained during the first order thermal decomposition of SO2Cl2 at a constant volume:
SO2Cl2(g) → SO2(g) + Cl2(g)
|
Experiment |
Time/s |
Total pressure/atm |
|
1 |
0 |
0.4 |
|
2 |
100 |
0.7 |
Calculate the rate constant.
(Given: log 4 = 0.6021, log 2 = 0.3010)
Q.8. Hydrogen peroxide, H2O2(aq) decomposes to H2O(l) and O2(g) in a reaction that is first order in H2O2 and has a rate constant k = 1.06 × 10−3 min−1.
(i) How long will it take for 15% of a sample of H2O2 to decompose?
(ii) How long will it take for 85% of the sample to decompose?
Q.9. The first order reaction take 100 minutes for completion of 60% of the reaction. Find the time when 90% of the reaction will be completed.
Q.10. For the reaction:
2NO(g) + Cl2(g) → 2NOCl(g)
The following data were collected. All the measurements were taken at 263 K.
|
Exp. No. |
Initial [NO] (M) |
Initial [Cl2] (M) |
Initial rate of disappearance of Cl2 (M/min.) |
|
1. |
0.15 |
0.15 |
0.60 |
|
2. |
0.15 |
0.30 |
1.20 |
|
3. |
0.30 |
0.15 |
2.40 |
|
4. |
0.25 |
0.25 |
? |
(i) Write the expression for rate law.
(ii) Calculate the value of rate constant and specify its units.
(iii) What is the initial rate of disappearance of Cl2 in exp. 4 ?
Q.11. A first order reaction is 15% completed in 20 minutes. How long will it take to complete 60% of the reaction?
Q.12. Nitrogen pentoxide decomposes according to equation:
2N2O5(g) → 4NO2(g) + O2(g)
This first order reaction was allowed to proceed at 40° C and the data below were collected:
|
[N2O5] (M) |
Time (min) |
|
0.400 |
0.00 |
|
0.289 |
20.0 |
|
0.209 |
40.0 |
|
0.151 |
60.0 |
|
0.109 |
80.0 |
(i) Calculate the rate constant. Include units with answer.
(ii) What will be the concentration of N2O5 after 100 minutes?
(iii) Calculate the initial rate of reaction.
(iv) After how many minutes will [N2O5] be equal to 0.350 M?
Q.13. A first order reaction has a rate constant value of 0.00510 min−1. If we begin with 0.10 M concentration of the reactant, how much of the reactant will remain after 3.0 hours?
Q.14. A reactant has a half−life of 10 minutes.
(i) Calculate the rate constant for the first order reaction.
(ii) What fraction of the reactant will be left after an hour of the reaction has occurred?
Q.15. TI1e decomposition of a compound is found to follow a first order rate law. If it takes 15 minutes for 20 per cent of original material to react, calculate
(i) the rate constant.
(ii) the time at which 10% of the original material remains unreacted.
Q.16. The half−life for a first order reaction is 5 × 104 s. What percentage of the initial reactant will react in 20 hours?
Q.17. A first order reaction is 20% complete in 5 minutes. Calculate the time taken for the reaction to be 60% complete.
Q.18. Define the Pseudo first order reaction
Q.19. For the hydrolysis of methyl acetate in aqueous solution, the following results were obtained:
|
t/s |
0 |
30 |
60 |
|
[CH3COOCH3] mol L−1 |
0.60 |
0.30 |
0.15 |
(i) Show that it follows pseudo first order reaction, as the concentration of water remains constant.
(ii) Calculate the average rate of reaction between the time interval 30 to 60 seconds.
(Given: log 2 = 0.3010, log 4 = 0.6021)
Q.20. In a pseudo first order hydrolysis of ester in water, the following results are obtained :
|
t in seconds |
0 |
30 |
60 |
90 |
|
[Ester]M |
0.55 |
0.31 |
0.17 |
0.085 |
(i) Calculate the average rate of reaction between the time interval 30 to 60 seconds.
(ii) Calculate the pseudo first order rate constant for the hydrolysis of ester.
Online Tuitions and Self-Study Courses for Grade 6 to 12 & JEE / NEET
