Chapter 6 – Combustion and Flame
Chapter Notes
A chemical process in which a substance reacts with the oxygen (of air) to give heat and light is called combustion.
Those substances which can burn are called combustible substances.
Some of the combustible substances are: Paper, Cloth (Fabrics), Straw (Dry grass), Cooking gas (LPG), CNG, Kerosene oil, Wood, Charcoal, Coal, Cow-dung cakes, Petrol, Diesel, Alcohol, Matchstick and Magnesium ribbon, etc. A combustible substance is also called a fuel.
Those substances which do not burn are called non-combustible substances.
Some of the non-combustible substances are: Stone, Glass, Cement, Bricks, Soil, Sand, Water, Iron nails, Copper objects and Asbestos, etc.
Some examples of combustion
(i) Combustion of Magnesium: If a magnesium ribbon is heated, it starts burning (or undergoes combustion). When a magnesium ribbon burns, it combines with the oxygen of air to form magnesium oxide and liberates heat and light.
Magnesium + Oxygen  Magnesium oxide + Heat + Light
Magnesium oxide + Heat + Light
(From air)
(ii) Combustion of Charcoal: Charcoal is mainly carbon. If we hold a piece of charcoal with a pair of tongs and heat it on the flame of a burner, it starts burning (or undergoes combustion). When charcoal burns, then the carbon of charcoal combines with the oxygen of air to form carbon dioxide. A lot of heat is produced in this combustion reaction but only a little light is produced (which makes the charcoal glow).
Carbon + Oxygen  Carbon dioxide + Heat + Light
Carbon dioxide + Heat + Light
(Charcoal) (From air)
Coal also contains a lot of carbon. So, coal also burns in air producing carbon dioxide, heat, and light.
Thus, charcoal and coal are combustible substances.
In both the examples of combustion given above, we find that oxygen is necessary for combustion (or burning) to take place.
(iii) Respiration is Combustion: Food is a fuel for our body. During respiration, the digested food (like glucose) is broken down by reaction with oxygen in the body cells to produce carbon dioxide, water, and heat energy. This heat energy is utilised by our body. Thus, respiration is a kind of slow combustion of food which takes place in the body to produce heat energy.
(iv) Rusting is Combustion: The rust is formed when iron slowly combines with the oxygen present in air (in the presence of moisture) to form iron oxide. The process of rusting of iron is an example of slow combustion. The rusting liberates very little heat but no light.
(v) Combustion on Sun: The sun produces heat and light. The heat and light produced in the sun are not due to ordinary combustion (which takes place in the presence of oxygen of air). In the sun, heat and light are produced due to nuclear reactions (in which hydrogen is converted into helium with the release of heat and light).
Conditions Necessary for Combustion
There are three conditions which are necessary for combustion to take place. These are:
1. Presence of a combustible substance (A substance which can burn)
The presence of a combustible substance is necessary for combustion to take place. So, when fire starts in a room, we remove all the combustible substances like wooden furniture, clothes, books and papers, etc., quickly from the room so that the fire may not spread due to the presence of a large number of combustible substances. A combustible substance is actually the food for fire.
2. Presence of a supporter of combustion (like air or oxygen)
The most common supporter of combustion which we have around us is air.
Activity to shows that air is necessary for combustion
Light a candle with a burning matchstick and fix it on a table. We will see that this candle keeps on burning. This uncovered candle keeps burning because it is getting continuous supply of fresh air from the surroundings. Let us now cover the burning candle with an inverted gas jar.
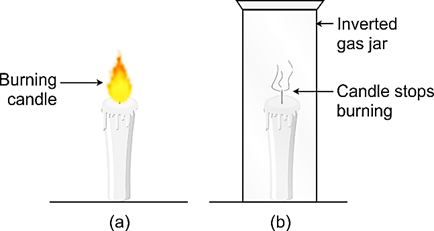
We will see that the candle stops burning after some time. In other words, the candle gets extinguished. The candle stops burning (or gets extinguished) because the supply of fresh air to the burning candle is cut off by the gas jar cover.
This observation shows that air is necessary for combustion (or burning) to take place.
Other examples:
(i) If burning charcoal is covered with a vessel, it stops burning after some time, that is, the charcoal fire gets extinguished after some time. Actually, when we cover the burning charcoal with a vessel, the supply of supporter of combustion (air) to the burning charcoal is cut off and hence the charcoal fire stops.
(ii) When the clothes of a person catch fire, the person is covered with a blanket to extinguish the fire. This is because when the burning clothes of a person are covered with a blanket, the supply of air to the burning clothes is cut off due to which the clothes stop burning the fire gets extinguished.
3. Heating the combustible substance to its ignition temperature
The lowest temperature at which a substance catches fire and starts burning, is called its ignition temperature. It is necessary to heat a combustible substance to its ignition temperature so that it may undergo combustion (or burn). The ignition temperature of paper is 233 °C. This means that a piece of paper has to be heated at least to a temperature of 233 °C so that it may catch fire and start burning. A combustible substance cannot catch fire (or burn) as long as its temperature is lower than its ignition temperature.
We usually apply a burning matchstick (or lighter) to make a substance burn. This burning matchstick supplies heat to raise the temperature of the substance to its ignition temperature and make it burn.
Some substances have low ignition temperatures whereas other substances have comparatively high ignition temperatures.
(i) Some of the substances having low ignition temperatures are: Paper, Splinters of wood, Dry grass (Straw), White phosphorus, Cloth (Fabrics), Alcohol, Kerosene, Petrol, LPG, CNG and Biogas. The lower the ignition temperature, the more easily the substance will catch fire. The substances which have very low ignition temperatures and can easily catch fire with a flame are called inflammable substances. Some of the examples of inflammable substances are: Petrol, Alcohol, Liquefied petroleum gas (LPG), Compressed natural gas (CNG) and Biogas. All these inflammable substances have very low ignition temperatures.
The fuels having very low ignition temperatures are very dangerous to use. For example, petrol has a much lower ignition temperature than that of kerosene. Due to its very low ignition temperature, a can full of petrol catches fire very easily on being lighted with a matchstick and burns explosively. This is why, petrol is not used in stoves. On the other hand, kerosene has a comparatively higher ignition temperature due to which it burns smoothly in a kerosene stove.
(ii) Some of the substances having high ignition temperatures are: Coal, Charcoal, Log of wood and Cow-dung cakes. The substances having high ignition temperatures catch fire with difficulty. The substances having high ignition temperatures burn only on strong heating. They cannot be burnt directly by a lighted matchstick.
How a matchstick is lighted
A matchstick is lighted by rubbing it on the rough surface provided on the side of the matchbox. The heat produced by friction raises the temperature of the chemicals present on the matchstick head to their ignition temperature. Due to this, the chemicals present on the head of the matchstick catch fire and the matchstick starts burning.

Kerosene oil and wood do not catch fire on their own at room temperature. This is because the ignition temperatures of kerosene oil and wood are higher than the room temperature. Now, if kerosene is heated a little (say, by a burning matchstick), it will catch fire easily. This is because kerosene oil has a comparatively low ignition temperature which is reached even on little heating. But if wood is heated a little, it does not catch fire. This is because wood has a much higher ignition temperature which is not reached by the little heat being supplied to it by a matchstick.
A matchstick can light a tiny splinter of wood but not a big log of wood. This is because a splinter of wood has a low ignition temperature. A burning matchstick can produce sufficient heat to reach the ignition temperature of the splinter of wood (which is low), therefore, a matchstick can light (or burn) a splinter of wood directly. The ignition temperature of a log of wood is high which cannot be reached by the small heat produced by a burning matchstick. So, a matchstick cannot light (or burn) a log of wood directly.
Coal has a high ignition temperature, so a coal fire cannot be started by using a lighted matchstick directly.
The forest fires occur during the hottest summer days. This happens because during extreme heat of summer, sometimes the ignition temperature of dry grass in the forest is reached, which makes the dry grass catch fire. From the burning grass, the fire spreads to bushes and trees, and very soon the whole forest is on fire. It is very difficult to control such forest fires.
Activity to show that Paper starts burning only on Ignition temperature
Take two paper cups.
Keep one paper cup empty but put about 50 ml of water in the other paper cup. Heat both the paper cups separately over a candle flame or a burner.

Empty paper cup catches fire easily and starts burning.
The paper cup containing water does not catch fire. The water in this paper cup becomes hot gradually. If we continue heating this paper cup, we can even boil the water in it without the paper cup catching fire.
Observations
(i) When we heat the empty paper cup over the burner, then the ignition temperature of paper is reached quickly. Due to this, the empty paper cup catches fire quickly and starts burning.
(ii) When we heat the paper cup containing water, then the heat supplied to the paper cup is transferred to water inside it by conduction. Due to the continuous transfer of heat from paper cup to water, the paper cup does not get heated too much and its ignition temperature is not reached. So, in the presence of water, the ignition temperature of paper cup is not reached, and hence the paper cup does not catch fire (or does not burn).
It is difficult to burn a heap of green leaves, but dry leaves catch fire easily because water does not allow the green leaves to get heated to their ignition temperature easily and makes the burning of green leaves difficult. On the other hand, since dry leaves do not contain water, they get heated to their ignition temperature easily and hence catch fire easily.
Earlier, a mixture of antimony trisulphide, potassium chlorate and white phosphorus with some glue and starch was applied to the head of a matchstick made of suitable wood.
These days, the head of matchstick (or safety match) contains only antimony trisulphide and potassium chlorate. The rough rubbing surface on the side of the matchbox has a coating of powdered glass and a little red phosphorus (Red phosphorus is much less dangerous than white phosphorus). When the matchstick is rubbed against the rough surface of matchbox, some red phosphorus is converted into white phosphorus. This white phosphorus immediately reacts with potassium chlorate in the matchstick head to produce sufficient heat to ignite antimony trisulphide and make the matchstick head burn.
How do We Control Fire?
Fire can be extinguished in three ways:
1. By removing the fuel (combustible substances)
2. By removing the heat (by cooling with water)
3. By cutting off the air supply to the burning substances (with carbon dioxide, etc.)
1. Remove the Fuel (or Combustible Substances)
A fuel (or combustible substance) is a food for fire. So, when fire starts in a room, then if possible, all the combustible substances like cooking gas cylinder, furniture, clothes, and books, etc., (which can burn easily) should be removed at once so that fire may not spread. If possible, electricity should be switched off.
Water is used to remove heat from a burning substance and to make it too cool to burn further. Water is the most common fire extinguisher for ordinary fires. Firemen extinguish the fire by throwing water under pressure on the burning things such as houses, factories, buildings, etc.
Water works as a fire extinguisher only when things like wood and paper, etc., are on fire.
The fire produced by burning oil and petrol (like fire in frying pan, oil tanks, petrol pumps and airports, etc.) cannot be extinguished by using water. This is because water is heavier than oil and petrol. So, when water is thrown over burning oil (or petrol), it (water) settles down. The oil (or petrol) floats on water and continues to burn. Thus, fires caused by burning oil (or petrol) cannot be extinguished by pouring water over it.
Also, the fires caused by electrical short-circuit in an electrical appliance or in electric wiring should not be extinguished by throwing water. This is because of the following reason: Ordinary water conducts electricity to some extent. So, when water is thrown over the burning electrical appliance (or burning electric wires), it can give electric shock to the persons involved in firefighting. Thus, water cannot be used to extinguish fires caused by electricity.
Many fires can be extinguished by cutting off air supply to the burning substances. The air supply to a burning substance can be cut off in a number of ways such as covering the burning substances with carbon dioxide, sand (or soil), a blanket or a damp cloth, etc.
The electrical fires are extinguished by using carbon dioxide gas fire extinguisher.
Carbon dioxide gas is denser than air and forms a layer around the burning substances. Carbon dioxide layer covers the fire like a blanket due to which fresh air cannot reach the burning substances. The burning substance does not get oxygen of air and hence stops burning.

Carbon dioxide used for extinguishing fire can be stored as a liquid at high pressure in cylinders (called fire extinguishers). When released from the cylinder, carbon dioxide expands enormously in volume and cools down. In this way, carbon dioxide not only forms a blanket around the burning substance, it also cools down the burning substance. This makes carbon dioxide an excellent fire extinguisher. Another way to obtain carbon dioxide for extinguishing a fire is to release a lot of dry powder of chemicals like sodium bicarbonate (baking soda) or potassium bicarbonate over the fire. The heat of fire decomposes these chemicals to produce carbon dioxide gas. And this carbon dioxide then extinguishes the fire.
A small fire can be extinguished by throwing sand (or soil) over it. For example, when sand is thrown over burning kerosene oil, the sand covers it like a blanket. The sand cuts off the air supply to the burning kerosene oil due to which the fire gets extinguished. If the clothes of a person working in the kitchen catch fire, the person is immediately covered with a blanket. When the burning clothes of a person are covered with a blanket, the supply of air to the burning clothes is cut off and hence the burning (or fire) stops.
The three important types of combustion are:
The combustion reaction in which a large amount of heat and light are produced in a short time is called rapid combustion. E.g. fire caught by the cooking gas, burning of kerosene oil in a kerosene stove and the burning of wax in a candle.
The combustion reaction which occurs on its own, without the help of any external heat source, is called spontaneous combustion. In spontaneous combustion, the substance suddenly bursts into flames and starts burning (even without being heated). Spontaneous combustion takes place at room temperature. The heat required for spontaneous combustion is produced inside the substance by its slow oxidation.
Spontaneous combustion is usually undergone by those substances which have quite low ignition temperatures.
If we keep a piece of white phosphorus in a china dish, we will see that it catches fire by itself and starts burning (without being heated). Hence it is an example of spontaneous combustion. The heat required to start this spontaneous combustion is produced internally by the slow oxidation of phosphorus in air.
The spontaneous combustion of coal dust has resulted in many disastrous fires in coal mines, leading to the death of many persons working in deep coal mines.
Forest fires can also be started by spontaneous combustion reactions. Sometimes, due to the heat of the sun or due to the spark of lightning from the sky, spontaneous combustion of straw and forest wood takes place leading to forest fires.
3. Explosive Combustion (or Explosion)
A very fast combustion reaction in which a large amount of heat, light and sound are produced, is called explosive combustion or explosion. A large amount of gases is released quickly in, an explosive combustion. It is the rapid expansion of these gases which causes a loud sound.
When a cracker is ignited with a burning matchstick, the chemicals present in it undergo a sudden combustion producing heat, light, and a large volume of gases. The gases produced are heated by the heat evolved in the reaction. The hot gases expand rapidly and cause an explosion producing a loud sound. Explosive combustion (or explosion) can also take place if pressure is applied on the cracker by hitting it hard.
A material which is burnt to produce heat is called a fuel. Some of the common fuels are: Wood, Coal, LPG (Cooking gas), Kerosene, Petrol, Diesel, Natural gas, and Biogas. A fuel is a very good source of heat energy.
The fuels which we use for various purposes can be in the form of a solid, a liquid or a gas. Thus, there are three types of fuels: Solid fuels, Liquid fuels, and Gaseous fuels.
(i) Some of the examples of solid fuels are: Wood, Charcoal, Coal, Coke, Agricultural wastes, and Cow-dung cakes (Uple).
(ii) The examples of liquid fuels are: Kerosene, Petrol, Diesel, and Alcohol (Ethanol).
(iii) The examples of gaseous fuels are: Natural gas, Petroleum gas, Biogas and Coal gas.
We use different types of fuels for various purposes at home, in industry and in transport for running automobiles. Some of the fuels used in homes are wood, charcoal, coal, LPG, and kerosene. Some of the fuels used in industry are coal and natural gas. The fuels used in running automobiles (or vehicles) are petrol, diesel and CNG.
Fuel Efficiency (Calorific value of Fuels)
Different fuels produce different amounts of heat on burning. The efficiency of a fuel is expressed in terms of its calorific value.
The amount of heat produced by the complete burning (or complete combustion) of 1 kilogram of a fuel is called its calorific value.
The calorific value of a fuel is expressed in the unit of 'kilojoules per kilogram' (which is written in short form as kJ/kg). The 'calorific value' of a fuel is also known as 'heat value'.
Calorific Values of Some Common Fuels
|
Fuel |
Calorific value |
|
1. Cow-dung cakes |
6000 to 8000 kJ /kg |
|
2. Wood |
17000 to 22000 kJ/kg |
|
3. Coal |
25000 to 33000 kJ /kg |
|
4. Biogas |
35000 to 40000 kJ /kg |
|
5. Petrol |
45000 kJ/kg |
|
6. Kerosene |
45000 kJ/kg |
|
7. Diesel |
45000 kJ/kg |
|
8. Methane |
50000 kJ/kg |
|
9. CNG (Compressed Natural Gas) |
50000 kJ/kg |
|
10. LPG (Liquefied Petroleum Gas) |
55000 kJ/kg |
|
11. Hydrogen gas |
150000 kJ /kg |
Calorific value of LPG is 55000 kJ/kg. It means that if 1 kilogram of LPG is burnt completely, then it will produce 55000 kilojoules of heat energy.
Characteristics of an Ideal Fuel (or Good Fuel)
(i) It has a high calorific value. That is, it produces a large amount of heat (per unit mass).
(ii) It burns easily in air at a moderate rate. That is, it burns neither too fast nor too slow.
(iii) It has a proper ignition temperature (which is neither very low nor very high).
(iv) It does not produce any harmful gases or leaves any residue after burning (which may pollute the environment).
(v) It is cheap, readily available, and easy to transport.
There is no fuel which can be considered to be an ideal fuel.
Flame is the 'blaze' of a fire. It is called 'jwala' or 'lapat' in Hindi.

Flames of Candle, Kerosene lamp and Gas burner
A flame is a region where combustion (or burning) of gaseous substances takes place. All the gases which undergo combustion (or burn) produce flame. But only those solid and liquid fuels which vaporize on being heated, burn with a flame. LPG and biogas are gases which undergo combustion, so LPG and biogas burn by producing a flame. Wax and camphor are solid substances which vaporise (or form gases) on heating, so wax and camphor burn with a flame. Similarly, kerosene oil and mustard oil are liquids which form vapours (or gases) on being heated, so kerosene oil and mustard oil also burn by producing flames.
Molten wax and kerosene oil rise through the wick, get vaporised during burning and form flames.
Charcoal is a solid fuel which does not vaporise on heating. So, charcoal does not burn by producing a flame. Charcoal only glows on combustion. Similarly, coal is a solid fuel which does not vaporise on heating. So, coal also does not burn by producing a flame. Coal just glows red on combustion.

Coal does not burn by producing a flame. It just glows.
When fuels burn, the type of flame produced depends on the proportion of oxygen (of air) which is available for burning the fuel (or for combustion of fuel).
(i) When the oxygen supply (or air supply) is insufficient, then the fuels burn incompletely producing mainly a yellow flame. The yellow flame is caused by the glow of hot unburnt carbon particles produced due to incomplete combustion of fuel. This yellow flame produces light, so it is said to be a luminous flame (or light-giving flame). When wax burns in the form of a candle, it burns with a yellow, luminous flame. Thus, the colour of candle flame is mainly yellow. When kerosene is burned in a lamp, it also burns with a yellow, luminous flame.

Wax candle burns by producing a yellow flame | LPG burns by producing a blue flame
(ii) When the oxygen supply is sufficient, then the fuels burn completely producing mainly a blue flame. This blue flame does not produce much light, so it is said to be a non-luminous flame (or non-light-giving flame). In LPG stove (or kitchen stove), the LPG burns with a blue flame (which is a non-luminous flame). The blue flame is produced when the complete combustion of a fuel takes place. Complete combustion of LPG takes place in the kitchen gas stove.
A flame consists of three zones (or three parts). These are: innermost zone, middle zone, and outer zone. The three zones of a flame have different colours and different temperatures.
Three zones of a flame of candle flame
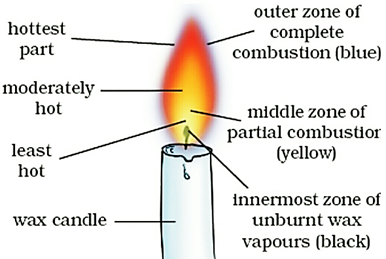
(i) The innermost zone of a flame is dark or black. The innermost zone of a flame consists of hot, unburnt vapours of the combustible material (say, wax vapours). The innermost zone is the least hot part of the flame. In other words, we can also say that the innermost zone (or dark zone) is the coldest part of the flame.
(ii) The middle zone of a flame is yellow. It is bright and luminous (light giving). The fuel vapours burn partially in the middle zone because there is not enough air for burning in this zone. The partial (or incomplete) burning of fuel in the middle zone produces carbon particles. These carbon particles become white hot and emit light. So, it is the glow of hot carbon particles which makes the middle zone of a flame luminous (or light-giving). These carbon particles then leave the flame as smoke and soot. The middle zone (or luminous zone) of a flame produces a moderate temperature. This zone is the major part of a candle flame.
(iii) The outer zone of a flame is blue. It is non-luminous zone which does not produce much light. In the outer zone of a flame, complete combustion of the fuel takes place because there is plenty of air around it. The outermost zone (or non-luminous zone) has the highest temperature in the flame. In other words, the outermost zone (or non-luminous zone) is the hottest part of the flame. The outermost zone of a flame is quite thin as compared to the middle zone.
Activity to Show that the Innermost Zone of a Candle Flame Consists of Unburnt Wax Vapours
Take a wax candle, fix it on a table and light it with a matchstick. Hold a thin glass tube with a pair of tongs and introduce one end of this glass tube in the innermost zone (dark zone or black zone) of the candle flame. Now bring a lighted matchstick near the other end of the glass tube. We will see a flame at this end of the glass tube. This is because the innermost zone (dark zone) of candle flame near the heated wick consists of unburnt wax vapours.

Some of these wax vapours enter the glass tube and come out from its other end. When we bring a lighted matchstick near this end of glass tube, the wax vapours coming out of it start burning, producing a flame. This activity shows that the innermost zone (dark zone or black zone) of a candle flame consists of unburnt wax vapours.
Activity to Show that Middle Zone of Candle Flame Consists of Unburnt Carbon Particles
Light a candle. Hold a clean glass plate with the help of a pair of tongs and introduce it in the middle zone (or luminous zone) of the candle flame. Hold the glass plate in this position for about 10 seconds. Then remove the glass plate from candle flame and observe it carefully. We will find that a blackish ring is formed on the glass plate.

This blackish ring is produced due to the deposition of unburnt carbon particles present in the luminous zone of the candle flame. This activity shows that the partial combustion of wax vapours in the middle zone produces unburnt carbon particles.
Activity to Show that the Outermost Zone (Non-Luminous Zone) of a Flame is Hottest
Take a long copper wire and hold its one end with a pair of tongs. Introduce the other end of copper wire just inside a burning candle flame so that it is in the outermost zone (non-luminous zone).

Keep the copper wire in this position for about 30 seconds. We will see that the part of copper wire which is in the outermost zone of the flame becomes red hot. This tells us that the outermost, non-luminous zone of a flame has a high temperature.
A kerosene oil lamp produces a flame exactly like the candle flame, consisting of the same three zones. Goldsmiths blow air with a blower to intensify a kerosene lamp flame for melting and moulding the pieces of gold and silver into desired shapes to make jewellery.
When air is blown through blowpipe into the flame, it helps in the combustion of unburnt fuel and hence makes the flame hotter.

Goldsmith blowing air into flame through a metallic blowpipe
Burning of Fuels Leads to Harmful Products
The important harmful effects produced by the burning of fuels are as follows:
1. The burning of fuels like wood, coal, and petroleum products (kerosene, petrol, diesel, etc.) releases unburnt carbon particles in the air. These fine carbon particles are dangerous pollutants which can cause respiratory diseases such as asthma.
2. Incomplete combustion of fuels (due to insufficient air) produces a very poisonous gas called carbon monoxide. Inhaling of carbon monoxide gas can kill a person. We should never sleep in a room with closed door and windows and having a coal fire burning inside. This is because when coal burns in an insufficient supply of air in the room (due to closed door and windows), then a lot of carbon monoxide gas is produced. When the persons sleeping in this room breathe in poisonous carbon monoxide gas, they may all die.
3. Burning of fuels releases carbon dioxide into air in the environment. Increased percentage of carbon dioxide in air is causing global warming. Carbon dioxide gas traps sun's heat rays by producing greenhouse effect. Global warming is the rise in temperature of earth's atmosphere caused by the excessive amounts of carbon dioxide in the air. Due to rise in the temperature of atmosphere, the ice in polar regions will melt very fast, producing a lot of water. This water may cause a rise in the sea-level leading to floods in coastal areas. The low-lying coastal areas may be completely submerged under water leading to the loss of life and property.
4. Burning of coal, petrol and diesel produces sulphur dioxide gas which goes into the air. Sulphur dioxide is an extremely suffocating and corrosive gas. It may damage our lungs. The burning of petrol and diesel in the engines of vehicles also releases nitrogen oxides into the air. Sulphur dioxide and nitrogen oxides produced by the burning of fuels dissolve in rainwater and form acids. The rainwater containing acids is called acid rain. Acid rain is very harmful for forests, aquatic animals, and buildings.
The use of petrol and diesel as fuels in automobiles (vehicles) is being replaced by CNG (Compressed Natural Gas). This is because when CNG burns, it produces very small amount of harmful products. CNG is a clean fuel because it burns without producing smoke. Since the burning of CNG produces much less harmful products and smoke, therefore, the use of CNG as fuel in automobiles has reduced air pollution in our cities.
(i) The burning of wood produces a lot of smoke which is very harmful for human beings. Smoke causes respiratory diseases.
(ii) Trees provide us with many useful substances. When trees are cut down to obtain wood for use as fuel, then all the useful substances which can be obtained from trees are lost.
(iii) The cutting down of trees to obtain fuel wood leads to deforestation which is very harmful for the environment. Wood has now been replaced by coal and other fuels such as LPG.
Advantages of LPG over wood:
(i) LPG has a much higher calorific value than wood, so it produces much more heat on burning than an equal mass of wood.
(ii) LPG burns without producing any smoke but burning of wood produces a lot of smoke.
(iii) LPG burns completely without leaving behind any solid residue but wood leaves behind a lot of ash on burning.
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
