EXAMIUM
CLASSES
www.examiumclasses.in
Class XII Chemistry
Term 2 [ Assignment 1 ]
Q.1. Why does physisorption decrease with the increase of temperature?
Q.2. Explain how the phenomenon of adsorption find application in the production of vacuum.
Q.3. Write one difference between solution and colloid.
Q.4. Write Werner’s postulates.
Q.5. Arrange the following compounds in increasing order of their basic strength:
(a) Aniline, p-nitroaniline and p-toluidine
(b) C6H5NH2, C6H5NHCH3, C6H5CH2NH2
Q.6. Write the IUPAC name of the following compounds:
(a) (H3CCH2)2NCH3
(b) H3CNHCH(CH3)2
(c) (H3C)2N–CH2CH3
Q.7. Limiting molar conductivity of an electrolyte cannot be determined experimentally. Why?
Q.8. Out of hexacyanomanganese(III) and hexachloridomanganese(III) which has maximum number of unpaired electrons. Why?
Q.9. Explain, cyclohexylamine is a stronger base than aniline. Draw resonating structures of aniline.
Q.10. For Freundlich adsorption isotherm, a plot of log (x/m) (y-axis) and log p (x-axis) gives a straight line. The intercept and slope for the line is 0.4771 and 2, respectively. What is the mass of gas, absorbed per gram of adsorbent if the initial pressure is 0.04 atm. (log 3 = 0.4771)
Q.11. What is meant by the following terms? Give an example of the reaction for their formation in each case.
(a) Cyanohydrin
(b) Acetal
(c) Semicarbazone
Q.12. How will you convert ethanal into the following compounds?
(a) Butane-1,3-diol
(b) But-2-enal
(c) But-2-enoic acid
Q.13. In following reaction sequence.
A: Blood red colour
B: Colourless
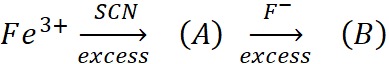
Identify (A) and (B). Give IUPAC name of (A). Find the spin only magnetic moment of (B).
Q.14. The conductivity of 0.20 M solution of KCl at 298 K is 0.0248 S cm–1. Calculate its molar conductivity.
Q.15. The resistance of a conductivity cell containing 0.001 M KCl solution at 298 K is 1500 Ω. What is the cell constant if conductivity of 0.001 M KCl solution at 298 K is 0.146 × 10–3 S cm–1?
Q.16. (a) What is lanthanoid contraction?
(b) Explain the following:
(i) Why is europium(II) more stable than cerium(II)?
(ii) Why is +3 oxidation state of gadolinium (Z = 64) and lutetium (Z = 71) especially stable?
Q.17. Give plausible explanation for each of the following :
(a) Cyclohexanone forms cyanohydrin in good yield but 2,2,6-trimethylcyclohexanone does not.
(b) There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
(c) During the preparation of esters from a carboxylic acid and an alcohol in the presence of an acid catalyst, the water or the ester should be removed as soon as it is formed.
Q.18. In a chemical reaction A + 2B → products, when concentration of A is doubled, rate of the reaction increases 4 times and when concentration of B alone is doubled rate continues to be the same. What is the order of the reaction?
Q.19. The rate constant of a reaction is found to be 3 × 10–3 mol–2 L2 sec–1. What is the order of the reaction ?
Q.20. In the reaction, A + 3B → 2C, what is the rate of formation of C ?
Q.21. Rate of a reaction can be expressed by following rate expression, Rate = k[A]2 [B], if concentration of A is increased by 3 times and concentration of B is increased by 2 times, how many times rate of reaction increases?
Q.22. The rate of a certain reaction is given by, rate = k[H+]n. The rate increases 100 times when the pH changes from 3 to 1. What is the order (n) of the reaction ?
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
