EXAMIUM
CLASSES
www.examiumclasses.in
Class XII Chemistry
Term 2 [ Assignment 1 ] Solutions
Q.1. Why does physisorption decrease with the increase of temperature?
Ans:
Physical adsorption of a gas by a solid is generally reversible. Thus,
Solid + Gas ⇌ Gas/Solid + Heat
Since the adsorption process is exothermic, the physical adsorption occurs readily at low temperature and decreases with increasing temperature (Le Chatelier’s principle).
Q.2. Explain how the phenomenon of adsorption find application in the production of vacuum.
Ans:
Production of vacuum : Adsorption can be applied to create condition of high vacuum. Vessel which has already been exhausted by vacuum pump is connected to a bulb containing charcoal. The remaining traces of air inspite of low pressure are adsorbed by the charcoal almost completely.
Q.3. Write one difference between solution and colloid.
Ans:
Solution : In true solution, the size of the particles is about 10–10 m.
Colloid : In a colloid, the size of the particles is in between 10–7 to 10–9 m.
Q.4. Write Werner’s postulates.
Ans:
The main postulates are :
(i) In coordination compounds metals show two types of linkages (valencies)-primary and secondary.
(ii) The primary valencies are normally ionisable and are satisfied by negative ions.
(iii) The secondary valencies are non ionisable. These are satisfied by neutral molecules or negative ions.
The secondary valency is equal to the coordination number and is fixed for a metal.
(iv) The ions/groups bound by the secondary linkages to the metal have characteristic spatial arrangements corresponding to different coordination numbers.
Q.5. Arrange the following compounds in increasing order of their basic strength:
(a) Aniline, p-nitroaniline and p-toluidine
(b) C6H5NH2, C6H5NHCH3, C6H5CH2NH2
Ans:
(a) p-Nitroaniline < Aniline < p-Toluidine
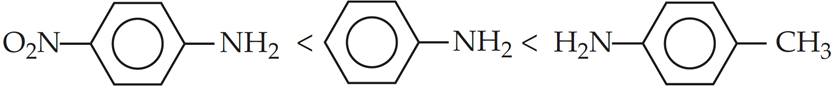
The availability of l.p. on N of p-nitroaniline is drastically reduced by presence of electron withdrawing –NO2 group on it.
In contrast, presence of electron releasing –CH3 group increases the electron density on N atom and improves basicity in p-toluidine.
(b) C6H5NH2(I) < C6H5NHCH3(II) < C6H5CH2NH2 (III)
Involvement of l.p. of N in resonance causes aniline to have low basicity. In II, the –Me group through its +I effect improves the electron density on N and therefore its basic strength increases. In III, the –NH2 is farther off from benzene ring and hence l.p. is localized on it and hence the basic strength is highest.
Q.6. Write the IUPAC name of the following compounds:
(a) (H3CCH2)2NCH3
(b) H3CNHCH(CH3)2
(c) (H3C)2N–CH2CH3
Ans:
(a) N-Ethyl-N-methylethanamine
(b) N-Methylpropan-2-amine (2° amine)
(c) N,N-Dimethylethanamine
Q.7. Limiting molar conductivity of an electrolyte cannot be determined experimentally. Why?
Ans:
In weak electrolyte, the conductivity of the solution increases very slowly with dilution of solution and goes on increasing up to infinity. Therefore, it cannot be measured experimentally.
Q.8. Out of hexacyanomanganese(III) and hexachloridomanganese(III) which has maximum number of unpaired electrons. Why?
Ans:
In [Mn(CN)6]3–, Mn is in +3 state so, it has configuration of 3d4.
Since CN– is a strong field ligand hence pairing of electrons in 3d-orbital takes place.

So, [Mn(CN)6]3– has two unpaired electrons. But in [MnCl6]3–, Cl– is a weak field ligand, so no pairing takes place and it has four unpaired electrons.
Q.9. Explain, cyclohexylamine is a stronger base than aniline. Draw resonating structures of aniline.
Ans:
Aniline is a weaker base than cyclohexylamine because of resonance.
Resonance structures of aniline are:

In case of cyclohexylamine there is no resonance.

Q.10. For Freundlich adsorption isotherm, a plot of log (x/m) (y-axis) and log p (x-axis) gives a straight line. The intercept and slope for the line is 0.4771 and 2, respectively. What is the mass of gas, absorbed per gram of adsorbent if the initial pressure is 0.04 atm. (log 3 = 0.4771)
Ans:
According to Freundlich adsorption isotherm,
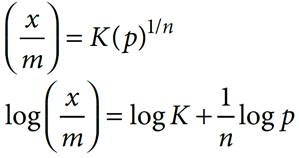
On comparing this with equation of straight line, we get,
Slope = ½ = 2
Intercept = log K = 0.4771
K = antilog (0.4771) = 3
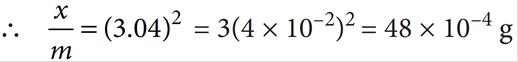
Q.11. What is meant by the following terms? Give an example of the reaction for their formation in each case.
(a) Cyanohydrin
(b) Acetal
(c) Semicarbazone
Ans:
(a) Cyanohydrin : Compounds formed when aldehydes or ketones react with hydrogen cyanide are called cyanohydrins.
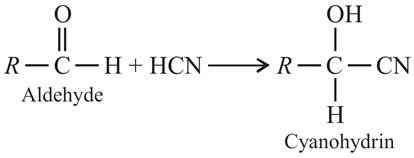
(b) Acetal : Acetals are the diethers which are formed by the addition of two molecules of alcohols to aldehydes in the presence of dry hydrogen chloride.
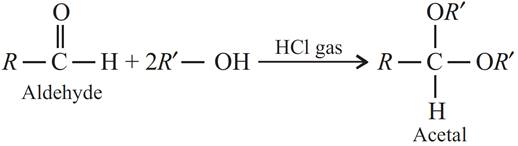
(c) Semicarbazone : Compounds formed when aldehydes or ketones react with semicarbazide are called semicarbazones.
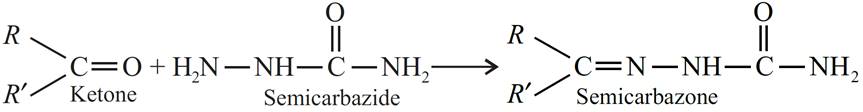
Q.12. How will you convert ethanal into the following compounds?
(a) Butane-1,3-diol
(b) But-2-enal
(c) But-2-enoic acid
Ans:
(a)
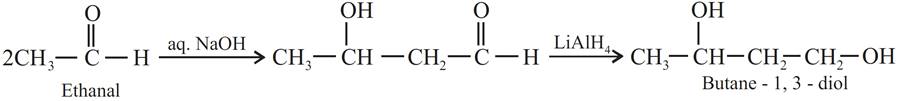
(b)
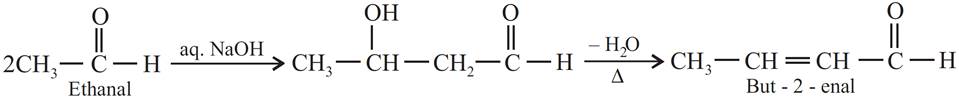
(c)
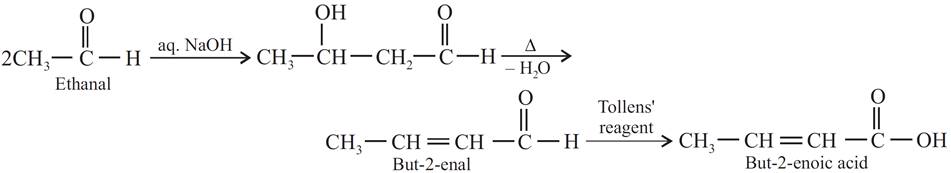
Q.13. In following reaction sequence.
A: Blood red colour
B: Colourless
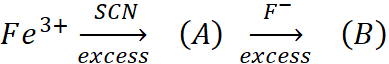
Identify (A) and (B). Give IUPAC name of (A). Find the spin only magnetic moment of (B).
Ans:
(A) = [Fe(SCN)(H2O)5]2+; (B) = [FeF6]3–
IUPAC name of A is : pentaaquathiocyanatoferrate(III) ion
Number of unpaired electrons in Fe3+ = 5
Spin magnetic moment of (B)
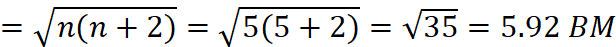
Q.14. The conductivity of 0.20 M solution of KCl at 298 K is 0.0248 S cm–1. Calculate its molar conductivity.
Ans:
Given, Conductivity, κ = 0.0248 S cm–1
Molarity, Cm = 0.20 M = 0.20 mol L–1
Using formula,
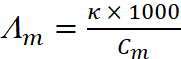
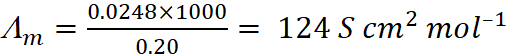
Q.15. The resistance of a conductivity cell containing 0.001 M KCl solution at 298 K is 1500 Ω. What is the cell constant if conductivity of 0.001 M KCl solution at 298 K is 0.146 × 10–3 S cm–1?
Ans:
Given, Cm = 0.001 M, R = 1500 Ω,
κ = 0.146 × 10–3 S cm–1, G* = ?
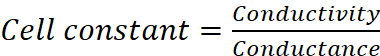
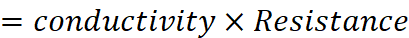
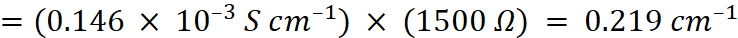
Q.16. (a) What is lanthanoid contraction?
(b) Explain the following:
(i) Why is europium(II) more stable than cerium(II)?
(ii) Why is +3 oxidation state of gadolinium (Z = 64) and lutetium (Z = 71) especially stable?
Ans:
(a) A unique feature of lanthanoids is the decrease in atomic and ionic radii from lanthanum to lutetium.
The gradual and steady decrease across the period is called lanthanoid contraction .
(b) (i) Europium (II) has electronic configuration [Xe] 4f7 5d0 while cerium (II) has electronic configuration [Xe] 4f1 5d1. In Eu2+, 4f subshell is half-filled and 5d-subshell is empty. Since half-filled and completely-filled electronic configurations are more stable, hence Eu2+ ions is more stable than Ce2+.
(ii) This is because gadolinium in +3 state has halffilled 4f-subshell (4f7) and lutetium in +3 state has completely filled 4f-subshell which are very stable configurations.
Q.17. Give plausible explanation for each of the following :
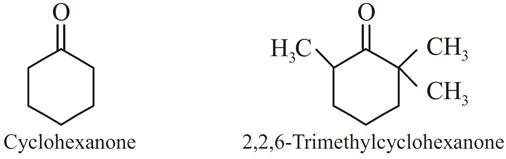
(a) Cyclohexanone forms cyanohydrin in good yield but 2,2,6-trimethylcyclohexanone does not.
(b) There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
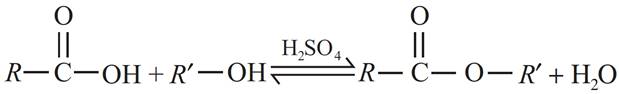
(c) During the preparation of esters from a carboxylic acid and an alcohol in the presence of an acid catalyst, the water or the ester should be removed as soon as it is formed.
Ans:
(a) Formation of cyanohydrin involves the nucleophilic attack of cyanide ions (CN–) at the carbonyl carbon. In cyclohexanone, reaction proceeds but in 2,2,6-trimethylcyclohexanone, the methyl groups cause steric hindrance and yields are poor.
(b) Although semicarbazide has to –NH2 groups but one of them which is directly attached to carbonyl group, is involved in resonance. So, this nitrogen is not able to act as an effective nucleophile.

So, it is the other —NH2 group which acts as nucleophile and is involved in condensation with aldehydes and ketones to give semicarbazone derivatives.
(c) During the preparation of ester from alcohol and carboxylic acid in presence of acid, the water or ester should be removed as fast as it is formed because it is a reversible reaction. If they are not removed, then the reverse reaction also starts and an equilibrium is established, so the overall yield of the ester will be low.
Q.18. In a chemical reaction A + 2B → products, when concentration of A is doubled, rate of the reaction increases 4 times and when concentration of B alone is doubled rate continues to be the same. What is the order of the reaction?
Ans:
Let the order of reaction w.r.t. A is x and w.r.t. B is y.
r1 = k[A]x [B]y ...(i)
r2 = k[2A]x [B]y ...(ii)
r3 = k[A]x [2B]y ...(iii)
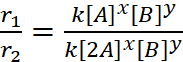
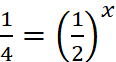
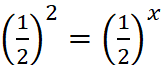
∴ x = 2
Similarly,
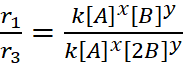
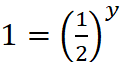
∴ y = 0
Hence the rate law equation is
Rate = k[A]2[B]0 ⇒ Order of reaction = 2
Q.19. The rate constant of a reaction is found to be 3 × 10–3 mol–2 L2 sec–1. What is the order of the reaction ?
Ans:
Unit of k for nth order = (mol L–1)1–n sec–1 ...(i)
Here, k = 3 × 10–3 mol–2 L2 sec–1
Unit of k = mol–2 L2 sec–1 ⇒ (mol L–1)–2 sec–1 ...(ii)
Comparing (i) and (ii) we get, 1 – n = –2 ⇒ n =3
Q.20. In the reaction, A + 3B → 2C, what is the rate of formation of C ?
Ans:

Q.21. Rate of a reaction can be expressed by following rate expression, Rate = k[A]2 [B], if concentration of A is increased by 3 times and concentration of B is increased by 2 times, how many times rate of reaction increases?
Ans:
Given, R1 = k[A]2 [B]
According to question, R2 = k[3A]2 [2B]
= k × 9 [A]2 × 2 [B] = 18 × k [A]2 [B] = 18 R1
Q.22. The rate of a certain reaction is given by, rate = k[H+]n. The rate increases 100 times when the pH changes from 3 to 1. What is the order (n) of the reaction ?
Ans:
Rate (r) = k[H+]n
When pH = 3 ; [H+] = 10–3
and when pH = 1 ; [H+] = 10–1
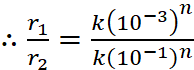
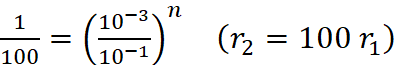
(10–2)1 = (10–2)n
Hence, n = 1
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
