Class 12 Chemistry
Magical Series Assignment – W4.AU.10
Find the Compounds
Q.1. An organic compound contains 69.77% carbon, 11.63% hydrogen and the rest is oxygen. The molecular mass of the compound is 86. It does not reduce Tollen's reagent but forms an addition compound with sodium hydrogen sulphite and gives a positive iodoform test. On vigorous oxidation it gives ethanoic and propanoic acids. Deduce the possible structure of the organic compound.
Q.2. A compound A (C2H6O) on oxidation by PCC gave B, which on treatment with aqueous alkali and subsequent heating furnished C. B on oxidation by KMnO4 forms a monobasic carboxylic acid with molar mass 60 g mol–1. Deduce the structures of A, B and C.
Q.3. An organic compound (A) on treatment with acetic acid in the presence of sulphuric acid produces an ester (B). (A) on mild oxidation gives (C). (C) with 50% KOH followed by acidification with dil. HCl generates (A) and (D). (D) with PCl5 followed by reaction with ammonia gives (E). (E) on dehydration produces hydrocyanic acid. Identify the compounds (A), (B), (C), (D), and (E).
Q.4. Compound A (C7H6Cl2) on treatment with NaOH solution gives another compound B (C7H6O). B, on oxidation gives an acid C (C7H6O2) which on treatment with a mixture of concentrated HNO3 and H2SO4 gives a compound D (C7H5NO4). B on treatment with conc. NaOH gives a compound E (C7H8O) and C6H5COONa. Deduce the structures of A, B, C, D and E.
Q.5. An organic compound A (C3H6O) is susceptible to oxidation and forms compound B (C3H8O) on reduction. B reacts with HBr to form the compound C. C with Mg forms Grignard reagent D which reacts with A to form a product which on hydrolysis gives E. Identify A to E.
Q.6. A ketone A (C4H8O), which undergoes a haloform reaction, gives compound B on reduction. B on heating with sulphuric acid gives a compound C which forms mono-ozonide D. D on hydrolysis in presence of Zn dust gives only acetaldehyde E. Identify A, B, C, D and E. Write the reactions involved.
Q.7. An organic compound with molecular formula C9H10O forms 2, 4-DNP derivative, reduces Tollen's reagent and undergoes Cannizzaro's 'reaction. On vigorous oxidation it gives benzene-1, 2- dicarboxylic acid. Identify the compound.
Q.8. An organic compound A, having the formula C3H8O on treatment with copper at 573 K, gives B. B does not reduce Fehling solution but gives a yellow precipitate of the compound C with I2/NaOH. Deduce the structures of A, B and C.
Q.9. An organic compound 'A' which has characteristic odour on treatment with NaOH forms two compounds 'B' and 'C'. Compound 'B' has the molecular formula C7H8O which on oxidation with CrO3 gives back compound 'A'. Compound 'C' is the sodium salt of the acid. 'C' when heated with soda lime yields an aromatic hydrocarbon 'D'. Deduce the structures of A, B, C and D.
Q.10. Compound (A), C6H12O2 on reduction with LiAlH4 yields two compounds (B) and (C). The compound (B) on oxidation gave (D) which on treatment with aqueous alkali and subsequent heating furnished (E). The latter on catalytic hydrogenation gave (C). The compound (D) on further oxidation gave CH3COOH. Deduce the structures of (A), (B), (C), (D) and (E).
Q.11. An organic compound (A), molecular formula C8H16O2, was hydrolysed with dilute sulphuric acid to give a carboxylic acid (B) and an alcohol (C). Oxidation of (C) with chromic acid produced (B). (C) on dehydration gives but-1-ene. Write chemical equations for the reactions involved.
Q.12. An organic compound (A) with molecular formula C8H8O forms an orange red precipitate with DNP reagent and gives yellow precipitate on heating with 12 and NaOH. It neither reduces Tollen's reagent nor Fehling reagent nor does it decolorize bromine water or Baeyer's reagent. On drastic oxidation with chromic acid, it gives a carboxylic acid. (B) having molecular formula C7H6O2. Identify the compounds (A) and (B) and explain the reactions involved.
Q.13. An organic compound (A) on treatment with ethyl alcohol gives a carboxylic acid (B) and compound (C). Hydrolysis of (C) under acidified conditions gives (B) and (D). Oxidation of (D) with KMnO4 also gives (B). (B) on heating with Ca(OH)2 gives (E) having molecular formula C3H6O. (E) does not give Tollen's test and does not reduce Fehling solution but forms a 2, 4-dinitrophenylhydrazone. Identify (A), (B), (C), (D) and (E).
Q.14. Two moles of an organic compound 'A' on treatment with a strong base gives two compounds 'B' and ' C '. Compound 'B' on dehydrogenation with Cu gives 'A' while acidification of 'C' yields carboxylic acid 'D' with molecular formula of CH2O2. Identity the compounds A, B, C and D and write all chemical reactions involved.
Answers
Ans.1. CH3CH2CH2COCH3
Ans.2. A: CH3CH2OH
B: CH3CHO
C: CH3CH=CHCHO
Ans.3. A: CH3OH
B: CH3COOCH3
C: HCHO
D: HCOOH
E: HCONH2 (methanamide)
Ans.4. A: C6H5CHCl2
B: C6H5CHO
C: C6H5COOH,
D: m-nitrobenzoic acid
E: C6H5CH2OH
Ans.5. A: CH3CH2CHO
B: CH3CH2CH2OH
C: CH3CH2CH2Br
D: CH3CH2CH2MgBr
E: CH3CH2CH(OH)CH2CH2CH3
Ans.6. A: CH3CH2COCH3
B: CH3CH2CH(OH)CH3
C: CH3CH=CHCH3
D: Ozonide
E: CH3CHO
Ans.7. 2-Ethyulbenzaldehyde
Ans.8. A: CH3CH(OH)CH3
B: CH3COCH3
C: CHI3
Ans.9. A: C6H5CHO
B: C6H5CH2OH
C: C6H5COONa
D: C6H6
Ans.10.
A: CH3CH2CH2COOCH2CH3
or CH3COOCH2CH2CH2CH3
B: CH3CH2OH
C: CH3CH2CH2CH2OH
D: CH3CHO
E: CH3CH=CHCHO (Crotonaldehyde)
Ans.11.
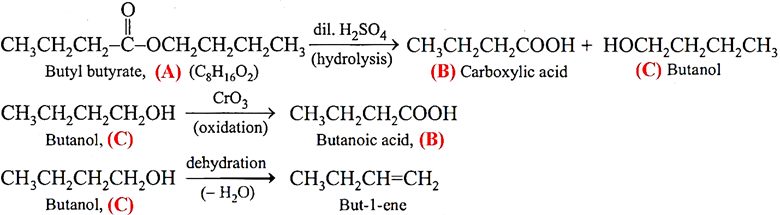
Ans.12. A: C6H5COCH3
B: C6H5COOH
Ans.13.
A: (CH3CO)2O (Acetic anhydride)
B: CH3COOH
C: CH3COOCH2CH3
D: CH3CH2OH
E: CH3COCH3
Ans.14. A: HCHO
B: CH3OH
C: HCOONa
D: HCOOH
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
