Class 12 Chemistry
Chapter 3 – Electrochemistry
Study Notes
Part – I
Electrolytes or Electrolytic conductors
These are substances which allow the electricity to pass through them in their molten states or in the form of their aqueous solutions and undergo chemical decomposition. For example, acids, bases and salts are electrolytes. The flow of electric current through an electrolytic solution is called electrolytic conduction. In this type of conduction, charge is carried by ions. Therefore, it is also called ionic conductance. Thus, the conduction will not occur unless the ions of the electrolyte are free to move. Therefore, these substances do not conduct electricity in the solid state.
Differences between Metallic and Electrolytic Conduction
|
Metallic conduction |
Electrolytic conduction |
|
1. Metallic conduction is carried by the movement of electrons. 2. It involves no change in the chemical properties of the conductor. 3. It does not involve the transfer of any matter. 4. Metallic conduction decreases with increase in temperature. |
Electrolytic conduction is carried by the movement of ions. It involves the decomposition of the electrolyte as a result of the chemical reaction It involves the transfer of matter as ions. Electrolytic conduction increases with increase in temperature. |
Strong electrolytes: The electrolytes which are almost completely dissociated into ions in solution are called strong electrolytes. For example, NaCl, KCl, HCl, NaOH, NH4NO3, etc.
Weak electrolytes: The electrolytes which do not ionise completely in solution are called weak electrolytes. For example, CH3COOH, H2CO3, H3BO3, HCN, HgCl2, ZnCl2, NH4OH, etc. Thus, in case of weak electrolytes, in equilibrium is established between the unionised electrolyte and the ions formed in solution. The extent of ionisation of a weak electrolyte is expressed in terms of degree of ionisation or degree of dissociation. It is defined as the fraction of total number of molecules of the electrolyte which ionise in the solution. It is generally denoted by alpha (α).
For strong electrolytes, α is almost equal to 1 and for weak electrolytes, it is always less than 1.
Factors Affecting Electrical Conductivity of Electrolytic Solutions
The conductivity of electrolytic (or ionic) solution depends upon the following factors:
(i) Nature of electrolyte. The conductance of an electrolyte depends upon the number of ions present in the solution. Therefore, the greater the number of ions in the solution, the greater is the conductance. The strong electrolytes dissociate almost completely into ions in solutions and therefore, their solutions have high conductance. On the other hand, weak electrolytes, dissociate to only small extents and give lesser number of ions. Therefore, the solutions of weak electrolytes have low conductance.
(ii) Nature of the solvent and its viscosity. Electrolytes ionize more in polar solvents. Therefore, greater the polarity of the solvent, larger is the ionization and hence greater. is the conductance. Similarly, greater is the viscosity of a solvent, lesser is its conductance.
(iii) Size of the ions produced and their solvation. If the ions are strongly solvated, their effective size will increase and hence their conductance will decrease.
(iv) Concentration of the electrolytic solution. Higher the concentration of the solution, less is the conductance. This is because, in a weak electrolyte, the ionization is less whereas in a strong electrolyte, the interionic attractions are large at higher concentrations. In general, the conductance of an electrolyte increases with decrease in concentration or increase in dilution.
With dilution, in case of weak electrolytes, ionization increases and hence conductance increases. In case of strong electrolytes, the interionic attractions decrease with dilution and therefore, conductance increases.
(v) Temperature. The conductivity of an electrolyte depends upon the temperature. With increase in temperature, the conductivity of an electrolyte increases due to decrease in interactions between ions.
When a voltage is applied to the electrodes dipped into an electrolytic solution, ions of the electrolyte move and, therefore, electric current flows through the electrolytic solution. The power of the electrolytes to conduct electric current is termed conductance or conductivity. Like metallic conductors, electrolytic solutions also obey Ohm's law.
Ohm's law
This law states that the current flowing through a conductor is directly proportional to the potential difference across it, i.e.,
I ∝
V or 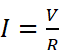 or V = IR
or V = IR
where I is the current strength (in amperes). V is the potential difference applied across the conductor (in volts) and R (in ohms, Ω) is the constant of proportionality and is known as resistance of the conductor.
In words, Ohm's law may be stated as the strength of current flowing through a conductor is directly proportional to the potential difference applied across the conductor and inversely proportional to the resistance of the conductor.
Resistance (R): It measures the obstruction to the flow of current. The resistance of any conductor is directly proportional to the length (l) and inversely proportional to the area of cross-section (a) so that
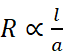 or
or
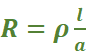
where ρ, (Greek, rho) is the constant of proportionality and is called resistivity or specific resistance.
Resistivity (𝛒): We know that resistance R is
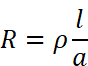
If l = 1 cm, a = 1 cm2, then R = ρ
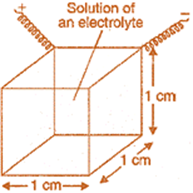
Thus, resistivity is defined as the resistance of a conductor of 1 cm length and having area of cross-section equal to 1 cm2.
In other words, resistivity or specific resistance is the resistance between opposite faces of one centimetre cube of the conductor. In terms of SI units, resistivity of a substance may be defined as its resistance when it is 1 m long and its area of cross section is 1 m2 i.e., resistance of one metre cube of the conductor.
Conductance (G): It is a measure of the ease with which the current flows through a conductor. It is expressed as 'G'. It is reciprocal of the resistance; i.e.,
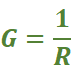
Conductivity (κ): The inverse of resistivity is called conductivity (or specific conductance). It is represented by the symbol, κ (Greek kappa). It may be defined as the conductance of a solution of 1 cm length and having 1 sq. cm as the area of cross-section. In other words, conductivity is the conductance of one centimetre cube of a solution of an electrolyte.
Thus, 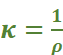
Molar Conductivity or Molar Conductance
Molar conductivity is defined as the conducting power of all the ions produced by dissolving one mole of an electrolyte in solution. It is denoted by Λm (lambda). Molar conductance is related to conductivity (κ) as:
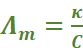 , where C is the molar concentration.
, where C is the molar concentration.
If C is in the units of molarity i.e. moles per litre (mol L-1), then Λm may be expressed as:
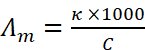 ,
where C is molarity (M).
,
where C is molarity (M).
Unit of Molar Conductivity is S cm2 mol-1.
1 S m2 mol-1 = 104 S cm2 mol-1
or 1 S cm2 mol-1 = 10-4 S m2 mol-1
Relation between Conductivity (κ) and Molar Conductivity (Λm)
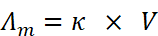
where V is the volume of the solution in cm3 containing one gram-mole of the electrolyte.
As 1 mole of electrolyte is present
in 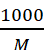 cm3
of solution, hence
cm3
of solution, hence
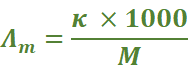
Measurement of conductance by Conductivity Cell
We know that accurate measurement of an unknown resistance can be performed on a Wheatstone bridge. However, for measuring the resistance of an ionic solution we face two problems. Firstly, passing direct current (DC) changes the composition of the solution. Secondly, a solution cannot be connected to the bridge like a metallic wire or other solid conductor.
The first difficulty is resolved by using an alternating current (AC) source of power. The second problem is solved by using a specially designed vessel called conductivity cell.
Two simple conductivity cells are shown in the figure below.
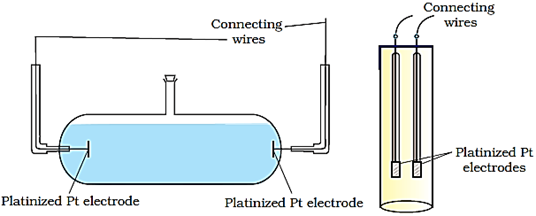
The electrodes of the cell are made up of platinum, coated with a thin layer of finely divided platinum called platinum black. This is done by the electrolysis of a solution of chloroplatinic acid. The coating of electrodes with platinum black reduces polarization effects.
Conductivity cell consists of two platinum electrodes coated with platinum black (finely divided metallic Pt is deposited on the electrodes electrochemically). These have area of cross section equal to ‘A’ and are separated by distance ‘l’. Therefore, solution confined between these electrodes is a column of length l and area of cross section A. The resistance of such a column of solution is then given by the equation:
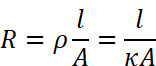
The quantity l/A is called cell constant denoted by the symbol, G*. It depends on the distance between the electrodes and their area of cross-section and has the dimension of length–1 and can be calculated if we know l and A. Measurement of l and A is not only inconvenient but also unreliable. The cell constant is usually determined by measuring the resistance of the cell containing a solution whose conductivity is already known.
The cell constant, G*, is then given by the equation:
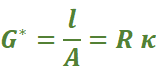
Once the cell constant is determined, we can use it for measuring the resistance or conductivity of any solution. The set up for the measurement of the resistance is shown in the figure below.
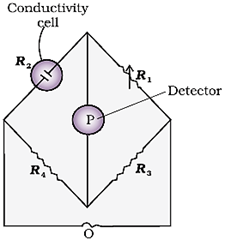
It consists of two resistances R3 and R4, a variable resistance R1 and the conductivity cell having the unknown resistance R2. The Wheatstone bridge is fed by an oscillator O (a source of AC power in the audio frequency range 550 to 5000 cycles per second). P is a suitable detector (a headphone or other electronic device) and the bridge is balanced when no current passes through the detector. Under these conditions:
Unknown resistance 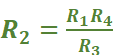
Once the cell constant and the resistance of the solution in the cell is determined, the conductivity of the solution is given by the equation:
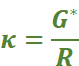
Variation of Conductivity and Molar Conductivity with Concentration
Conductivity always decreases with decrease in concentration both, for weak and strong electrolytes.
This can be explained by the fact that the number of ions per unit volume that carry the current in a solution decreases on dilution.
The conductivity of a solution at any given concentration is the conductance of one unit volume of solution kept between two platinum electrodes with unit area of cross section and at a distance of unit length. This is clear from the equation:
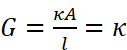 (both
A and l are unity in their appropriate units in m or cm)
(both
A and l are unity in their appropriate units in m or cm)
Molar conductivity of a solution at a given concentration is the conductance of the volume V of solution containing one mole of electrolyte kept between two electrodes with area of cross section A and distance of unit length. Therefore,
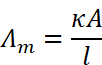
Since l = 1 and A = V (volume containing 1 mole of electrolyte)
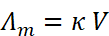
Molar conductivity increases with
decrease in concentration. This is because the total volume, V, of solution
containing one mole of electrolyte also increases. It has been found that
decrease in κ on dilution of a solution is more than compensated by
increase in its volume. Physically, it means that at a given concentration,
Λm can be defined as the conductance of the electrolytic
solution kept between the electrodes of a conductivity cell at unit distance
but having area of cross section large enough to accommodate sufficient volume
of solution that contains one mole of the electrolyte. When concentration
approaches zero, the molar conductivity is known as limiting molar
conductivity and is represented by the symbol 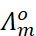 .
The variation in Λm with concentration is different for strong
and weak electrolytes (shown in the figure below)
.
The variation in Λm with concentration is different for strong
and weak electrolytes (shown in the figure below)
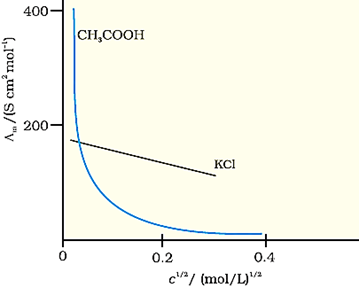
Strong Electrolytes
For strong electrolytes, Λm increases slowly with dilution and can be represented by the equation:
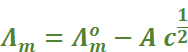
It can be seen that if we plot Λm
against 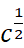 ,
we obtain a straight line with intercept equal to
,
we obtain a straight line with intercept equal to 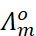 and
slope equal to ‘–A’. The value of the constant ‘A’ for a given solvent and
temperature depends on the type of electrolyte i.e., the charges on the cation
and anion produced on the dissociation of the electrolyte in the solution.
and
slope equal to ‘–A’. The value of the constant ‘A’ for a given solvent and
temperature depends on the type of electrolyte i.e., the charges on the cation
and anion produced on the dissociation of the electrolyte in the solution.
Thus, NaCl, CaCl2, MgSO4 are known as 1–1, 2–1 and 2–2 electrolytes respectively. All electrolytes of a particular type have the same value for ‘A’.
Kohlrausch Law
Kohlrausch examined 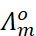 values
for a number of strong electrolytes and observed certain regularities. He noted
that the difference in
values
for a number of strong electrolytes and observed certain regularities. He noted
that the difference in 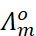 of
the electrolytes NaX and KX for any X is nearly constant. For example, at 298
K:
of
the electrolytes NaX and KX for any X is nearly constant. For example, at 298
K:
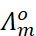 (KCl)
–
(KCl)
– 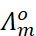 (NaCl)
=
(NaCl)
= 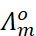 (KBr)
–
(KBr)
– 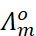 (NaBr)
=
(NaBr)
= 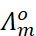 (KI)
–
(KI)
– 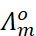 (NaI)
Y 23.4 S cm2 mol–1
(NaI)
Y 23.4 S cm2 mol–1
and similarly, it was found that
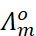 (NaBr)
–
(NaBr)
– 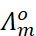 (NaCl)
=
(NaCl)
= 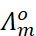 (KBr)
–
(KBr)
– 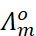 (KCl)
Y 1.8 S cm2 mol–1
(KCl)
Y 1.8 S cm2 mol–1
On the basis of the above observations he enunciated Kohlrausch law of independent migration of ions. The law states that limiting molar conductivity of an electrolyte can be represented as the sum of the individual contributions of the anion and cation of the electrolyte.
Thus, if 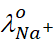 and
and
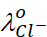 are
limiting molar conductivity of the sodium and chloride ions respectively, then
the limiting molar conductivity for sodium chloride is given by the equation:
are
limiting molar conductivity of the sodium and chloride ions respectively, then
the limiting molar conductivity for sodium chloride is given by the equation:
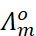 (NaCl)
=
(NaCl)
= 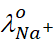 +
+
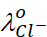
In general, if an electrolyte on dissociation gives ν+ cations and ν– anions then its limiting molar conductivity is given by:
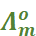 =
= 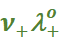 +
+ 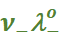
Here, 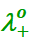 and
and
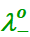 are
the limiting molar conductivities of the cation and
anion respectively.
are
the limiting molar conductivities of the cation and
anion respectively.
Weak Electrolytes
Weak electrolytes like acetic acid
have lower degree of dissociation at higher concentrations and hence for such
electrolytes, the change in Λm with dilution is due to increase
in the degree of dissociation and consequently the number of ions in total volume
of solution that contains 1 mol of electrolyte. In such cases Λm
increases steeply on dilution, especially near lower concentrations. Therefore,
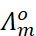 cannot
be obtained by extrapolation of Λm to zero concentration. At
infinite dilution (i.e., concentration c → zero) electrolyte dissociates
completely (α =1), but at such low concentration the conductivity of the
solution is so low that it cannot be measured accurately. Therefore,
cannot
be obtained by extrapolation of Λm to zero concentration. At
infinite dilution (i.e., concentration c → zero) electrolyte dissociates
completely (α =1), but at such low concentration the conductivity of the
solution is so low that it cannot be measured accurately. Therefore, 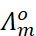 for
weak electrolytes is obtained by using Kohlrausch law of independent migration
of ions. At any concentration c, if a is the degree of dissociation then it can
be approximated to the ratio of molar conductivity Λm at the
concentration c to limiting molar conductivity,
for
weak electrolytes is obtained by using Kohlrausch law of independent migration
of ions. At any concentration c, if a is the degree of dissociation then it can
be approximated to the ratio of molar conductivity Λm at the
concentration c to limiting molar conductivity, 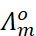 .
Thus, we have:
.
Thus, we have:
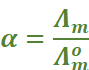
We know that for a weak electrolyte like acetic acid
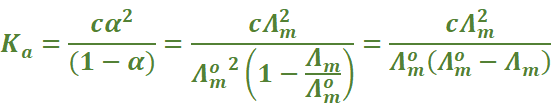
Part – II
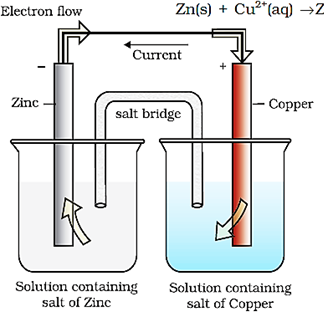
Electrochemical Cells
Daniell cell depicted by the reaction given below converts the chemical energy liberated during the redox reaction to electrical energy and has an electrical potential equal to 1.1 V when concentration of Zn2+ and Cu2+ ions is unity (1 mol dm–3).
Zn(s) + Cu2+(aq) → Zn2+(aq) + Cu(s)
Such a device is called a galvanic or a voltaic cell.
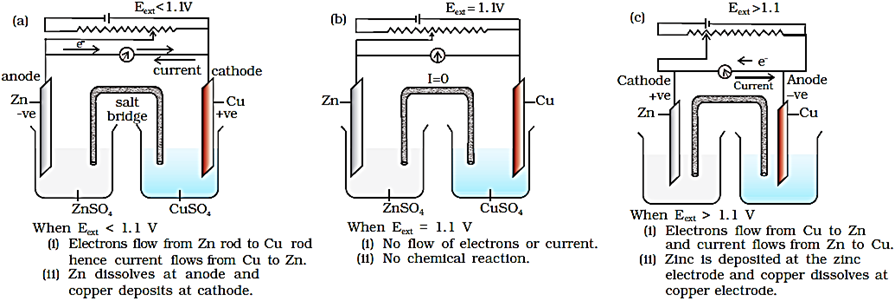
If an external opposite potential is applied in the galvanic cell and increased slowly beyond 1.1 V then, the reaction occurs in the opposite direction. It now functions as an electrolytic cell, a device for using electrical energy to carry non-spontaneous chemical reactions.
Galvanic Cells
Daniell cell discussed earlier is one Galvanic cell in which the following redox reaction occurs.
Zn(s) + Cu2+(aq) → Zn2+(aq) + Cu(s)
This reaction is a combination of two half reactions whose addition gives the overall cell reaction:
(i) Cu2+ + 2e– → Cu(s) (reduction half reaction)
(ii) Zn(s) → Zn2+ + 2e– (oxidation half reaction)
These reactions occur in two different portions of the Daniell cell.
The reduction half reaction occurs on the copper electrode while the oxidation half reaction occurs on the zinc electrode. These two portions of the cell are also called half-cells or redox couples. The copper electrode may be called the reduction half-cell and the zinc electrode, the oxidation half-cell.
In a galvanic cell, the half-cell in which oxidation takes place is called anode and it has a negative potential with respect to the solution. The other half-cell in which reduction takes place is called cathode and it has a positive potential with respect to the solution.
The potential difference between the two electrodes of a galvanic cell is called the cell potential and is measured in volts. It is called the cell electromotive force (emf) of the cell when no current is drawn through the cell.
It is now an accepted convention that we keep the anode on the left and the cathode on the right while representing the galvanic cell.
A galvanic cell is represented by putting a vertical line between metal and electrolyte solution and putting a double vertical line between the two electrolytes connected by a salt bridge.
Under this convention the emf of the cell is positive and is given by the potential of the half-cell on the right-hand side minus the potential of the half-cell on the left-hand side i.e.,
Ecell = Eright – Eleft
This is illustrated by the following example:
Cell reaction:
Cu(s) + 2Ag+(aq) → Cu2+(aq) + 2 Ag(s)
Half-cell reactions:
Cathode (reduction): 2Ag+(aq) + 2e– → 2Ag(s)
Anode (oxidation): Cu(s) → Cu2+(aq) + 2e–
It can be seen that the sum of half-reactions leads to overall reaction in the cell and that silver electrode acts as a cathode and copper electrode acts as an anode. The cell can be represented as:
Cu(s)|Cu2+(aq) || Ag+(aq)|Ag(s)
and we have Ecell = Eright – Eleft = EAg+|Ag – ECu2+|Cu
Measurement of Electrode Potential
The potential of individual half-cell cannot be measured. We can measure only the difference between the two half-cell potentials that gives the emf of the cell. If we arbitrarily choose the potential of one electrode (half-cell) then, that of the other can be determined with respect to this.
According to convention, a half-cell called standard hydrogen electrode (SHE) represented by Pt(s) | H2(g) | H+(aq), is assigned a zero potential at all temperatures corresponding to the reaction
H+ (aq) + e–
→  H2(g)
H2(g)
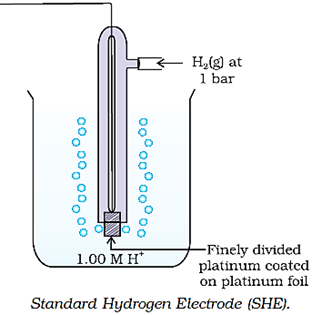
The standard hydrogen electrode consists of a platinum electrode coated with platinum black. The electrode is dipped in an acidic solution and pure hydrogen gas is bubbled through it. The concentration of both the reduced and oxidised forms of hydrogen is maintained at unity. This implies that the pressure of hydrogen gas is one bar and the concentration of hydrogen ion in the solution is one molar.
At 298 K the emf of the cell,
standard hydrogen electrode || second half-cell constructed by taking standard
hydrogen electrode as anode (reference half-cell) and the other half-cell as
cathode, gives the reduction potential of the other half-cell. If the
concentrations of the oxidised and the reduced forms of the species in the
right-hand half-cell are unity, then the cell potential is equal to standard
electrode potential, 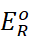 of
the given half-cell.
of
the given half-cell.
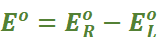
As 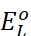 for
standard hydrogen electrode is zero.
for
standard hydrogen electrode is zero.
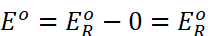
The measured emf of the cell:
Pt(s) | H2(g, 1 bar) | H+ (aq, 1 M) || Cu2+ (aq, 1 M) | Cu
is 0.34 V and it is also the value for the standard electrode potential of the half-cell corresponding to the reaction:
Cu2+ (aq, 1M) + 2 e– → Cu(s)
Similarly, the measured emf of the cell:
Pt(s) | H2(g, 1 bar) | H+ (aq, 1 M) || Zn2+ (aq, 1M) | Zn
is –0.76 V corresponding to the standard electrode potential of the half-cell reaction:
Zn2+ (aq, 1 M) + 2e– → Zn(s)
The positive value of the standard electrode potential in the first case indicates that Cu2+ ions get reduced more easily than H+ ions.
The reverse process cannot occur, that is, hydrogen ions cannot oxidise Cu (or alternatively we can say that hydrogen gas can reduce copper ion) under the standard conditions described above. Thus, Cu does not dissolve in HCl. In nitric acid it is oxidised by nitrate ion and not by hydrogen ion.
The negative value of the standard electrode potential in the second case indicates that hydrogen ions can oxidise zinc (or zinc can reduce hydrogen ions).
In view of this convention, the half reaction for the Daniell cell can be written as:
Left electrode: Zn(s) → Zn2+ (aq, 1 M) + 2 e–
Right electrode: Cu2+ (aq, 1 M) + 2 e– → Cu(s)
The overall reaction of the cell is the sum of above two reactions and we obtain the equation:
Zn(s) + Cu2+ (aq) → Zn2+ (aq) + Cu(s)
emf of the cell = 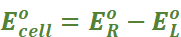
= 0.34 V – (– 0.76) V = 1.10 V
Sometimes metals like platinum or gold are used as inert electrodes. They do not participate in the reaction but provide their surface for oxidation or reduction reactions and for the conduction of electrons.
For example, Pt is used in the following half-cells:
Hydrogen electrode: Pt(s) | H2(g)| H+(aq)
With half-cell reaction: H+ (aq) + e– → ½ H2(g)
Bromine electrode: Pt(s) | Br2(aq) | Br–(aq)
With half-cell reaction: ½ Br2(aq) + e– → Br–(aq)
If the standard electrode potential of an electrode is positive then its reduced form is more stable compared to hydrogen gas.
If the standard electrode potential is negative then hydrogen gas is more stable than the reduced form of the species.
It can be seen that the standard electrode potential for fluorine is the highest, indicating that fluorine gas (F2) has the maximum tendency to get reduced to fluoride ions (F–) and therefore fluorine gas is the strongest oxidising agent and fluoride ion is the weakest reducing agent.
Lithium has the lowest electrode potential indicating that lithium ion is the weakest oxidising agent while lithium metal is the most powerful reducing agent in an aqueous solution.
As we go from top to bottom in the reduction potential series the standard electrode potential decreases and with this, decreases the oxidising power of the species on the left and increases the reducing power of the species on the right-hand side of the reaction.
We have assumed in the previous section that the concentration of all the species involved in the electrode reaction is unity. This need not be always true. Nernst showed that for the electrode reaction:
Mn+(aq) + ne– → M(s)
the electrode potential at any concentration measured with respect to standard hydrogen electrode can be represented by:
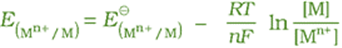
but concentration of solid M is taken as unity and we have
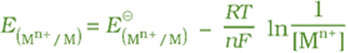
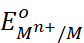 has
already been defined and a table has been prepared for the same. R is gas
constant (8.314 J K–1 mol–1), F is Faraday constant
(96487 C mol–1), T is temperature in kelvin and [Mn+] is
the concentration of the species, Mn+.
has
already been defined and a table has been prepared for the same. R is gas
constant (8.314 J K–1 mol–1), F is Faraday constant
(96487 C mol–1), T is temperature in kelvin and [Mn+] is
the concentration of the species, Mn+.
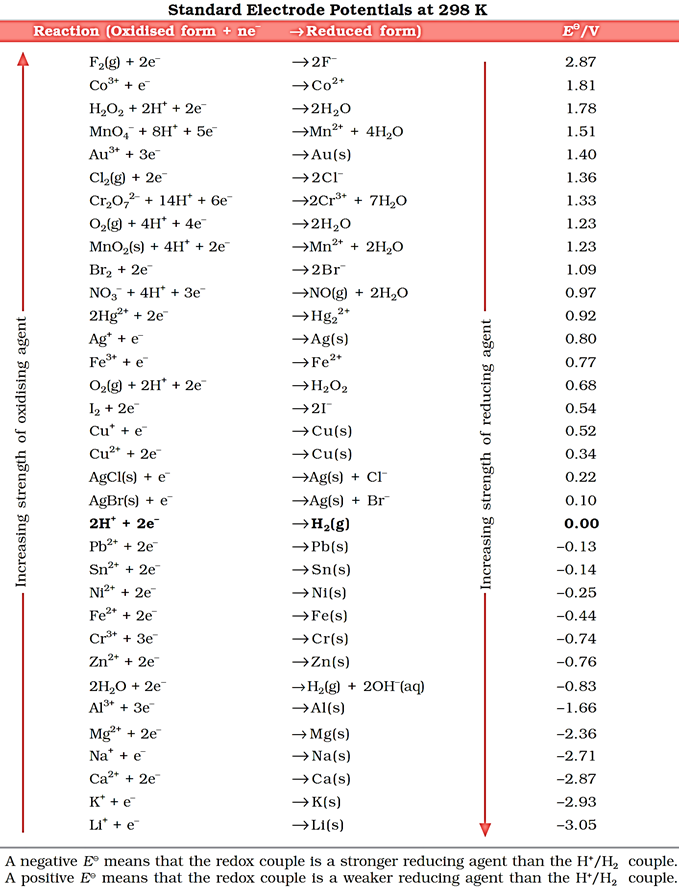
In Daniell cell, the electrode potential for any given concentration of Cu2+ and Zn2+ ions,
For Cathode:
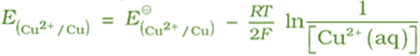
For Anode:
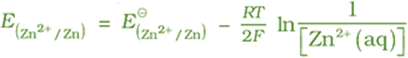
The cell potential:

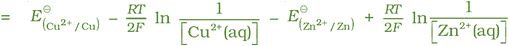
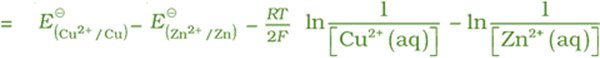
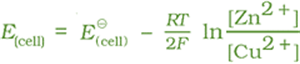
It can be seen that E(cell) depends on the concentration of both Cu2+ and Zn2+ ions. It increases with increase in the concentration of Cu2+ ions and decrease in the concentration of Zn2+ ions.
With logarithm at base 10 and substituting the values of R, F and T = 298 K, it reduces to
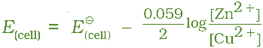
We should use the same number of electrons (n) for both the electrodes and thus for the cell
Ni(s) | Ni2+(aq) || Ag+(aq) | Ag
The cell reaction is Ni(s) + 2Ag+(aq) → Ni2+(aq) + 2Ag(s)
The Nernst equation can be written as
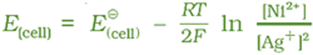
and for a general electrochemical reaction of the type:

Nernst equation can be written as:
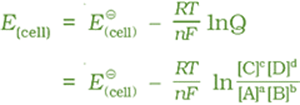
Equilibrium Constant from Nernst Equation
If the circuit in Daniell cell is closed then we note that following the reaction takes place
Zn(s) + Cu2+(aq) → Zn2+(aq) + Cu(s)
and concentration of Zn2+ keeps on increasing and concentration of Cu2+ keeps on decreasing.
At the same time voltage of the cell as read on the voltmeter keeps on decreasing. After some time, we shall note that there is no change in the concentration of Cu2+ and Zn2+ ions and at the same time, voltmeter gives zero reading. This indicates that equilibrium has been attained. In this situation the Nernst equation may be written as:
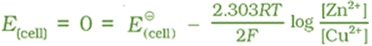
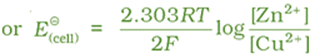
But at equilibrium,
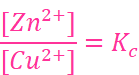
and at T = 298 K the above equation can be written as
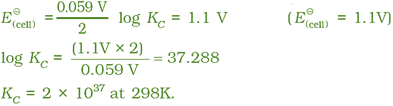
In general,
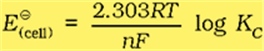
Electrochemical Cell and Gibbs Energy of the Reaction
Electrical work done in one second is equal to electrical potential multiplied by total charge passed. If we want to obtain maximum work from a galvanic cell then charge has to be passed reversibly. The reversible work done by a galvanic cell is equal to decrease in its Gibbs energy and therefore, if the emf of the cell is E and nF is the amount of charge passed and ∆rG is the Gibbs energy of the reaction, then
∆rG = – nFE(cell)
It may be remembered that E(cell) is an intensive parameter but ∆rG is an extensive thermodynamic property and the value depends on n.
Thus, if we write the reaction
Zn(s) + Cu2+(aq) → Zn2+(aq) + Cu(s)
∆rG = – 2FE(cell)
but when we write the reaction
2 Zn (s) + 2 Cu2+(aq) → 2 Zn2+(aq) + 2Cu(s)
∆rG = – 4FE(cell)
If the concentration
of all the reacting species is unity, then 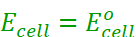 and
we have
and
we have
∆rGo
= – nF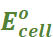
Thus, from the measurement of 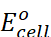 we
can obtain an important thermodynamic quantity,
we
can obtain an important thermodynamic quantity, 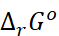 ,
standard Gibbs energy of the reaction.
,
standard Gibbs energy of the reaction.
From the latter we can calculate equilibrium constant by the equation:
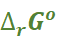 = –RT ln K.
= –RT ln K.
Part – III
Electrolytic Cells and Electrolysis
In an electrolytic cell external source of voltage is used to bring about a chemical reaction.
One of the simplest electrolytic cells consists of two copper strips dipping in an aqueous solution of copper sulphate.
If a DC voltage is applied to the two electrodes, then Cu2+ ions discharge at the cathode (negatively charged) and the following reaction takes place:
Cu2+(aq) + 2e– → Cu (s)
Copper metal is deposited on the cathode. At the anode, copper is converted into Cu2+ ions by the reaction:
Cu(s) → Cu2+(s) + 2e–
Thus, copper is dissolved (oxidised) at anode and deposited (reduced) at cathode. This is the basis for an industrial process in which impure copper is converted into copper of high purity.
The impure copper is made an anode that dissolves on passing current and pure copper is deposited at the cathode.
Faraday’s Laws of Electrolysis
First Law: The amount of chemical reaction which occurs at any electrode during electrolysis by a current is proportional to the quantity of electricity passed through the electrolyte (solution or melt).
Second Law: The amounts of different substances liberated by the same quantity of electricity passing through the electrolytic solution are proportional to their chemical equivalent weights (Atomic Mass of Metal ÷ Number of electrons required to reduce the cation).
Quantity of electricity Q, passed is given by
Q = It
Q is in coulombs when I is in ampere and t is in second.
The amount of electricity (or charge) required for oxidation or reduction depends on the stoichiometry of the electrode reaction. For example, in the reaction:
Ag+(aq) + e– → Ag(s)
One mole of the electron is required for the reduction of one mole of silver ions.
We know that charge on one electron is equal to 1.6021 × 10–19 C.
Therefore, the charge on one mole of electrons:
= NA × 1.6021 × 10–19 C
= 6.02 × 1023 mol–1 × 1.6021 × 10–19 C
= 96487 C mol–1
This quantity of electricity is called Faraday and is represented by the symbol F.
For approximate calculations we use 1F Y 96500 C mol–1.
For the electrode reactions:
Mg2+(l) + 2e– → Mg(s)
Al3+(l) + 3e– → Al(s) ,
it is obvious that one mole of Mg2+ and Al3+ require 2 mol of electrons (2F) and 3 mol of electrons (3F) respectively.
The products of electrolysis may be different for reactive and inert electrodes. The products of electrolysis depend on the different oxidising and reducing species present in the electrolytic cell and their standard electrode potentials.
For example, if we use molten NaCl, the products of electrolysis are sodium metal and Cl2 gas. Here we have only one cation (Na+) which is reduced at the cathode (Na+ + e– → Na) and one anion (Cl–) which is oxidised at the anode (Cl– → ½Cl2 + e–). During the electrolysis of aqueous sodium chloride solution, the products are NaOH, Cl2 and H2. In this case besides Na+ and Cl– ions we also have H+ and OH– ions along with the solvent molecules, H2O.
At the cathode there is competition between the following reduction reactions:
Na+(aq) + e–
→ Na(s) 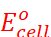 = – 2.71 V
= – 2.71 V
H+(aq) + e–
→ ½ H2(g) 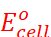 = 0.00 V
= 0.00 V
The reaction with higher value of Eo is preferred and therefore, the reaction at the cathode during electrolysis is:
H+(aq) + e– → ½ H2(g)
but H+(aq) is produced by the dissociation of H2O, i.e.,
H2O(l) → H+(aq) + OH–(aq)
Therefore, the net reaction at the cathode may be written as the sum of these two reactions and we have
H2O(l) + e– → ½H2(g) + OH–
At the anode the following oxidation reactions are possible:
Cl–(aq) → ½ Cl2(g)
+ e– 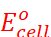 = 1.36 V
= 1.36 V
2H2O(l) → O2(g)
+ 4H+(aq) + 4e– 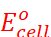 = 1.23 V
= 1.23 V
The reaction at anode with lower value of Eo is preferred and therefore, water should get oxidised in preference to Cl–(aq). However, on account of overpotential of oxygen,
Cl–(aq) → ½ Cl2(g) + e– reaction is preferred. Thus, the net reactions may be summarised as:
NaCl (aq) 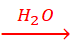 Na+(aq) + Cl– (aq)
Na+(aq) + Cl– (aq)
Cathode: H2O(l) + e– → ½ H2(g) + OH– (aq)
Anode: Cl–(aq) → ½ Cl2(g) + e–
Net reaction:
NaCl(aq) + H2O(l) → Na+(aq) + OH–(aq) + ½H2(g) + ½Cl2(g)
The standard electrode potentials are replaced by electrode potentials given by Nernst equation to take into account the concentration effects. During the electrolysis of sulphuric acid, the following processes are possible at the anode:
2H2O(l) → O2(g)
+ 4H+(aq) + 4e– 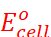 = +1.23 V ……… (i)
= +1.23 V ……… (i)
2SO42–(aq)
→ S2O82–(aq) + 2e– 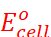 = 1.96 V ……… (ii)
= 1.96 V ……… (ii)
For dilute sulphuric acid, reaction (i) is preferred but at higher concentrations of H2SO4, reaction (ii) is preferred.
Any battery or cell that we use as a source of electrical energy is basically a galvanic cell where the chemical energy of the redox reaction is converted into electrical energy.
There are mainly two types of batteries.
(i) Primary Batteries
(ii) Secondary Batteries
In the primary batteries, the reaction occurs only once and after use over a period of time battery becomes dead and cannot be reused again.
The most familiar example of this type is the dry cell (known as Leclanche cell after its discoverer) which is used commonly in our transistors and clocks.
consists of a zinc container that also acts as anode and the cathode is a carbon (graphite) rod surrounded by powdered manganese dioxide and carbon. The space between the electrodes is filled by a moist paste of ammonium chloride (NH4Cl) and zinc chloride (ZnCl2). The electrode reactions are complex, but they can be written approximately as follows:
Anode: Zn(s) → Zn2+ + 2e–
Cathode: MnO2 + NH4+ + e– → MnO(OH) + NH3
In the reaction at cathode, manganese is reduced from the +4 oxidation state to the +3 state. Ammonia produced in the reaction forms a complex with Zn2+ to give [Zn (NH3)4]2+. The cell has a potential of nearly 1.5 V.
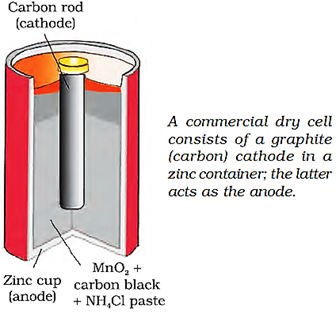
It is suitable for low current devices like hearing aids, watches, etc. consists of zinc – mercury amalgam as anode and a paste of HgO and carbon as the cathode.
The electrolyte is a paste of KOH and ZnO. The electrode reactions for the cell are given below:
Anode: Zn(Hg) + 2OH– → ZnO(s) + H2O + 2e–
Cathode: HgO + H2O + 2e– → Hg(l) + 2OH–
The overall reaction is represented by
Zn(Hg) + HgO(s) → ZnO(s) + Hg(l)
The cell potential is approximately 1.35 V and remains constant during its life as the overall reaction does not involve any ion in solution whose concentration can change during its life time.
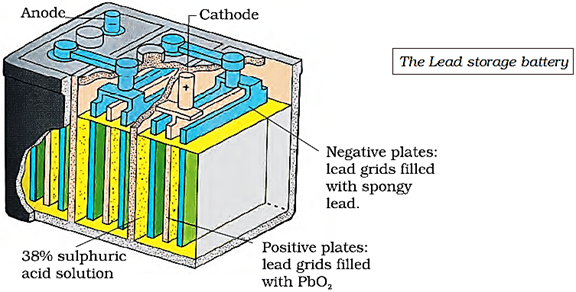
A secondary cell after use can be recharged by passing current through it in the opposite direction so that it can be used again. A good secondary cell can undergo a large number of discharging and charging cycles. The most important secondary cell is the lead storage battery, commonly used in automobiles and invertors.
It consists of a lead anode and a grid of lead packed with lead dioxide (PbO2) as cathode. A 38% solution of sulphuric acid is used as an electrolyte. The cell reactions are given below:
Anode: Pb(s) + SO42–(aq) → PbSO4(s) + 2e–
Cathode: PbO2(s) + SO42–(aq) + 4H+(aq) + 2e– → PbSO4(s) + 2H2O(l)
i.e., overall cell reaction consisting of cathode and anode reactions is:
Pb(s) + PbO2(s) + 2H2SO4(aq) → 2PbSO4(s) + 2H2O(l)
On charging the battery the reaction is reversed and PbSO4(s) on anode and cathode is converted into Pb and PbO2, respectively.
Nickel-Cadmium cell has longer life than the lead storage cell but is more expensive.
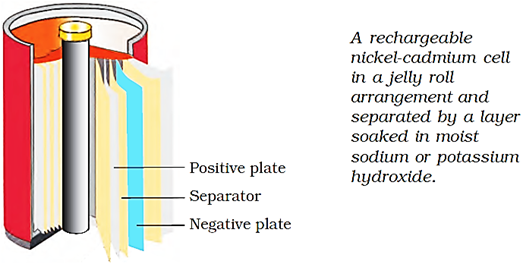
The overall reaction during discharge is:
Cd(s) + 2Ni(OH)3(s) → CdO(s) + 2Ni(OH)2(s) + H2O(l)
It is possible to make Galvanic cells in which reactants are fed continuously to the electrodes and products are removed continuously from the electrolyte compartment. Galvanic cells that are designed to convert the energy of combustion of fuels like hydrogen, methane, methanol, etc. directly into electrical energy are called fuel cells.
One of the most successful fuel cells uses the reaction of hydrogen with oxygen to form water. The cell was used for providing electrical power in the Apollo space programme. Water vapours produced during the reaction were condensed and added to the drinking water supply.
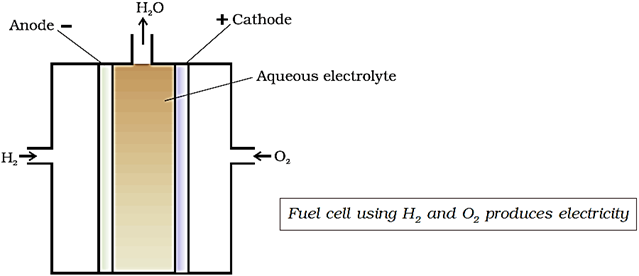
In the cell, hydrogen and oxygen are bubbled through porous carbon electrodes into concentrated aqueous sodium hydroxide solution. Catalysts like finely divided platinum or palladium metal are incorporated into the electrodes for increasing the rate of electrode reactions. The electrode reactions are given below:
Cathode: O2(g) + 2H2O(l) + 4e– → 4OH–(aq)
Anode: 2H2(g) + 4OH–(aq) → 4H2O(l) + 4e–
Overall reaction being:
2H2(g) + O2(g) → 2H2O(l)
The cell runs continuously as long as the reactants are supplied.
Fuel cells produce electricity with an efficiency of about 70 % compared to thermal plants whose efficiency is about 40%.
In corrosion, a metal is oxidised by loss of electrons to oxygen and formation of oxides.
Corrosion of iron (commonly known as rusting) occurs in presence of water and air.
The chemistry of corrosion is quite complex but it may be considered essentially as an electrochemical phenomenon.
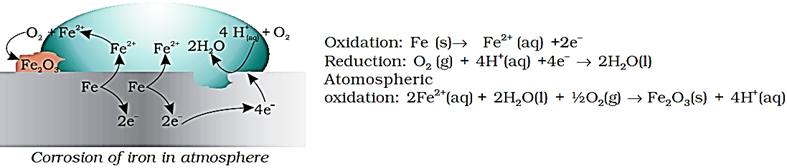
At a particular spot of an object made of iron, oxidation takes place and that spot behaves as anode and we can write the reaction
Anode: 2Fe(s) → 2Fe2+
+ 4e– 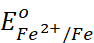 =
– 0.44 V
=
– 0.44 V
Electrons released at anodic spot move through the metal and go to another spot on the metal and reduce oxygen in the presence of H+ (which is believed to be available from H2CO3 formed due to dissolution of carbon dioxide from air into water. Hydrogen ion in water may also be available due to dissolution of other acidic oxides from the atmosphere). This spot behaves as cathode with the reaction
Cathode: O2(g) + 4H+(aq)
+ 4e– → 2H2O(l) 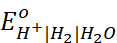 =1.23
V
=1.23
V
The overall reaction being:
2Fe(s) + O2(g) + 4H+(aq)
→ 2Fe2+(aq) + 2H2O(l) 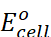 =1.67
V
=1.67
V
The ferrous ions are further oxidised by atmospheric oxygen to ferric ions which come out as rust in the form of hydrated ferric oxide (Fe2O3.xH2O) and with further production of hydrogen ions.
One of the simplest methods of preventing corrosion is to prevent the surface of the metallic object to come in contact with atmosphere. This can be done by covering the surface with paint or by some chemicals (e.g. bisphenol).
Another simple method is to cover the surface by other metals (Sn, Zn, etc.) that are inert or react to save the object. An electrochemical method is to provide a sacrificial electrode of another metal (like Mg, Zn, etc.) which corrodes itself but saves the object.
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
