Q.1. Given the data at 25° C:
Ag + I− → AgI + e− ; Eo = 0.152 V
Ag → Ag+ + e− ; Eo = − 0.800 V
(2.303 RT/F = 0.059 V)
What is the value of log Ksp for AgI?
Q.2. Resistance of a conductivity cell filled with a solution of an electrolyte of concentration 0.1 M is 100 W. The conductivity of this solution is 1.29 S m−1. Resistance of the same cell when filled with 0.2 M of the same solution is 520 W. Calculate the molar conductivity of 0.2 M solution of the electrolyte.
Q.3. The cell Zn | Zn2+ (1 M) || Cu2+
(1 M) | Cu (Eocell = 1.10 V) was allowed to be completely
discharged at 298 K. Calculate the relative concentration of Zn2+ to
Cu2+ , 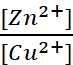 .
.
Q.4. Given Eo(Cr3+ | Cr) = − 0.72 V and Eo(Fe2+ | Fe) = − 0.42 V.
Find the potential for the cell Cr | Cr3+(0.1M) || Fe2+(0.01 M) | Fe.
Q.5. Given 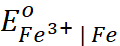 = − 0.036 V ;
= − 0.036 V ; 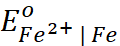 = − 0.439 V. Calculate the value of standard electrode
potential for the change, Fe3+ + e− → Fe2+.
= − 0.439 V. Calculate the value of standard electrode
potential for the change, Fe3+ + e− → Fe2+.
Q.6. The Gibbs energy for the decomposition of Al2O3 at 500°C is as follows:
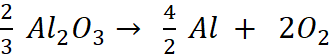
Calculate the minimum potential difference needed for electrolytic reduction of Al2O3 at 500°C.
Q.7. The reduction potential of hydrogen half−cell will be negative if:
(a) p(H2) = 2 atm and [H+] = 1.0 M
(b) p(H2) = 2 atm and [H+] = 2.0 M
(c) p(H2) = 1 atm and [H+] = 2.0 M
(d) p(H2) = 1 atm and [H+] = 1.0 M
Q.8. Resistance of 0.2 M solution of an electrolyte is 50 W. The specific conductance of the solution is 1.3S m−1. If resistance of the 0.4 M solution of the same electrolyte is 260 W, calculate its molar conductivity.
Q.9. The standard reduction potentials for Zn2+ | Zn, Ni2+ | Ni, and Fe2+ | Fe are −0.76,−0.23 and −0.44 V respectively.
The reaction X + Y2+ → X2+ + Y will be spontaneous when
(a) X = Ni, Y = Zn
(b) X = Fe, Y = Zn
(c) X = Zn, Y = Ni
(d) X = Ni, Y = Fe
Q.10. Given 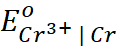 = −0.74 V;
= −0.74 V; 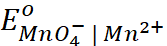 = = 1.51 V
= = 1.51 V
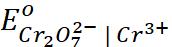 = 1.33 V;
= 1.33 V; 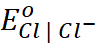 = 1.36 V
= 1.36 V
Based on the data given above, strongest oxidising agent will be
(a) MnO4−
(b) Cl−
(c) Cr3+
(d) Mn2+
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
