A solution is simply a homogeneous mixture of two or more substances.
Solutions can be solid, liquid and gaseous.
Alloy is an example of a solid solution, while air is a gaseous solution.
A mixture is called solution when it has homogeneity at the particle level.
Examples of solutions
|
Solutions |
Solutes |
Solvents |
|
1. Saltwater |
Salt |
Water |
|
2. Solution of iodine in alcohol |
Iodine |
Alcohol |
|
3. Vinegar |
Acetic acid |
Water |
|
4. Soda water |
Carbon dioxide |
Water |
|
5. Air |
Other gases (present in relatively smaller amounts) |
Nitrogen (present in the largest amount) |
|
Types of Solution |
Solute |
Solvent |
Examples |
|
Gaseous solutions |
Gas |
Gas |
Mixture of O2 and N2 |
|
Liquid |
Gas |
Chloroform mixed with N2 Gas |
|
|
|
Solid |
Gas |
Camphor in nitrogen gas |
|
Liquid Solutions |
Gas |
Liquid |
Oxygen dissolved in water |
|
|
Liquid |
Liquid |
Ethanol dissolved in water |
|
|
solid |
Liquid |
Glucose dissolved in water |
|
Solid Solutions |
Gas |
solid |
Solution of H2 and Pd |
|
|
Liquid |
solid |
Amalgam of Hg with Na |
|
|
Solid |
solid |
Copper dissolved with Gold (Alloy) |
Properties of Solutions
· They are homogeneous mixtures of solutes and solvents.
· The solute particles in a solution are extremely small in size. They are less than 1 nm (10−9 m) in diameter.
· The solute particles are not visible to the naked eye.
· As a result of the small size of the solute particles, a solution does not scatter a beam of light passing through it.
· Being small in size, the solute particles get dissolved in the solvent. Hence, the solute cannot be separated from the solvent by filtration.
· The solute particles do not settle down when left undisturbed.
Vapour Pressure
Like a gas, a liquid exerts pressure of its own due to evaporation. This pressure is known as the vapour pressure of the liquid.
Ways to represent Concentration
(i) Mass percentage (w/w):

(ii) Volume percentage (V/V):

(iii) Mass by volume percentage (w/V): Another unit which is commonly used in medicine and pharmacy is mass by volume percentage. It is the mass of solute dissolved in 100 mL of the solution.
(iv) Parts per million:
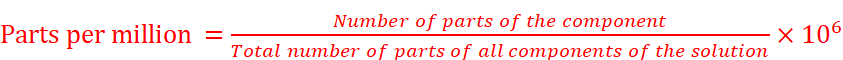
(v) Mole fraction:
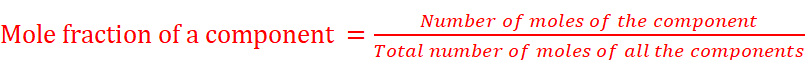
For example, in a binary mixture, if the number of moles of A and B are nA and nB respectively, the mole fraction of A will be
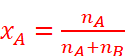
(vi) Molarity: Molarity (M) is defined as number of moles of solute dissolved in one litre (or one cubic decimetre) of solution,

(vii) Molality: Molality (m) is defined as the number of moles of the solute per kilogram (kg) of the solvent and is expressed as:
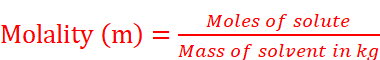
· Solubility of a substance is the maximum amount of the substance that can be dissolved in a specified amount of a solvent at a given temperature.
· Factors on which solubility depends:
· Nature of solute and solvent
· Temperature
· Pressure
Solubility of a Solid in a Liquid
When a solute is dissolved in a solvent, the following dynamic equilibrium is established.
Solute
+ Solvent  Solution
Solution
This equilibrium follows Le Chatelier’s Principle.
· Effect of temperature:
According to Le Chatelier’s principle, in a nearly saturated solution, if the dissolution process is
· Endothermic (ΔsolH > 0), then the solubility will increase with the increase in temperature
· Exothermic (ΔsolH < 0), then the solubility will decrease with the increase in temperature
· Effect of pressure:
· Pressure has no significant effect on the solubility of solids in liquids.
· Reason − Solids and liquids are negligibly affected by pressure as they are highly incompressible.
Solubility of a Gas in a Liquid
· The solubility of gases in liquids increases with the increase in pressure.
Henry’s law
The solubility of a gas in a liquid is directly proportional to the pressure of the gas.
Or
The partial pressure of a gas in vapour phase (p) is directly proportional to the mole fraction of the gas (x) in the solution, i.e.,
p = KH x
Where, KH = Henry’s law constant
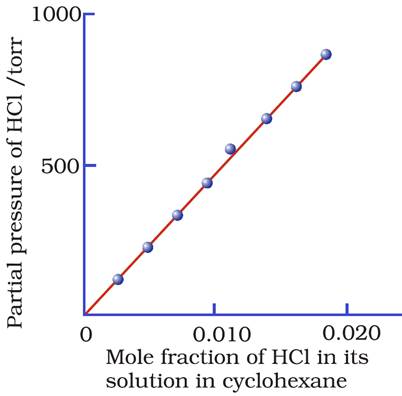
The plot of the partial pressure of HCl vs. its mole fraction in a solution of cyclohexane is given below.
Some important applications of Henry’s law
(i) Bottles are sealed under high pressure to increase the solubility of CO2 in soft drinks and soda water.
(ii) Henry’s law states that the solubility of gas increases with an increase in pressure. Therefore, when a scuba diver dives into the sea, the increased sea pressure causes the nitrogen present in air to dissolve in his blood in great amounts. As a result, when he comes back to the surface, the solubility of nitrogen again decreases and the dissolved gas is released, leading to the formation of nitrogen bubbles in the blood. This results in the blockage of capillaries and leads to a medical condition known as ‘bends’, which are painful and dangerous to life. Hence, the oxygen tanks used by scuba divers are filled with air and diluted with helium to avoid bends.
(iii) The concentration of oxygen is low in the blood and tissues of climbers or people living at high altitudes. This is because at high altitudes, the partial pressure of oxygen is less than that at ground level. Low blood oxygen causes climbers to become weak and prevents them from thinking clearly. These are symptoms of ‘anoxia’.
· Effect of temperature
Solubility of gases in liquids decreases with the increase in temperature.
Vapour Pressure of Liquid Solutions
· Let p1, p2 = Partial vapour pressure of two volatile components 1 and 2 of a mixture
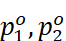 =
Vapour pressure of pure components 1 and 2
=
Vapour pressure of pure components 1 and 2
x1, x2 = Mole fractions of the components 1 and 2
ptotal = Total vapour pressure of the mixture
For a solution of volatile liquids, the partial vapour pressure of each component in the solution is directly proportional to its mole fraction.
p1 ∝ x1
That is, for component 1,
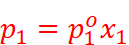
For component 2,
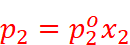
According to Dalton’s law of partial pressures,
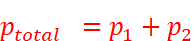
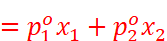
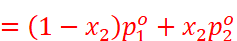

· From the above equation, it can be concluded that:
· ptotal can be related to mole fraction of any one component.
· ptotal varies linearly with x2.
·
Depending upon 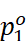 and
and
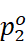 ,
, 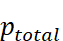 increases
or decreases with the increase of x1.
increases
or decreases with the increase of x1.
· The plot of vapour pressure and mole fraction of an ideal solution at constant temperature is shown below.
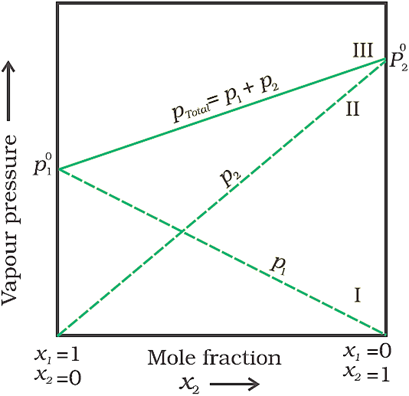
Here,
maximum value of 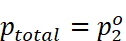 .
.
Minimum
value of 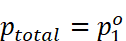 .
.
Here, 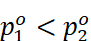 because
it is assumed that component 1 is less volatile than component 2.
because
it is assumed that component 1 is less volatile than component 2.
· The component of vapour phase in equilibrium with the solution is determined by the partial pressures of the components.
Let y1, y2 = Mole fractions of the components 1 and 2 respectively in the vapour phase.
According to Dalton’s law of partial pressures,
p1 = y1 ptotal
p2 = y2 ptotal
In general, we can write
pi = yi ptotal
Raoult’s Law as a Special Case of Henry’s Law
·
According to Raoult’s law, the vapour pressure of a volatile
component in a given solution is 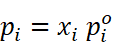 .
.
· According to Henry’s law, the partial vapour pressure of a gas (the component is so volatile that it exists as gas) in a liquid is
p = KH x
·
It can be observed that in both the equations, the partial vapour
pressure of the volatile component varies directly with its mole fraction. Only
the proportionality constants KH and 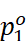 are
different. Thus, Raoult’s law becomes a special case of Henry’s law in
which KH is equal to
are
different. Thus, Raoult’s law becomes a special case of Henry’s law in
which KH is equal to 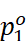 .
.
Vapour Pressure of Solutions of Solids in Liquids
· When a non-volatile solute is added to a solvent, the vapour pressure of the liquid decreases.
· Reason: The number of solvent molecules on the surface decreases and as a result, number of solvent molecules escaping from the surface decreases.
· Raoult’s law in general form: For any solution, the partial vapour pressure of each volatile component in the solution is directly proportional to its mole fraction.
· Let us take a binary solution made by dissolving a non-volatile solute in a solvent. Since the solute is non-volatile, only the solvent molecules contribute to vapour pressure.
Let 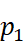 =
Vapour pressure of the solvent
=
Vapour pressure of the solvent
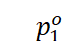 =
Vapour pressure of the solvent in pure state
=
Vapour pressure of the solvent in pure state
x1 = Mole fraction of the solvent
Then, according to Raoult’s law,
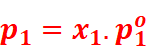
Here, the proportionality constant is equal to the vapour pressure of the solvent in pure state.
The plot of vapour pressure vs. mole fraction of the solvent, which is linear, is shown below.
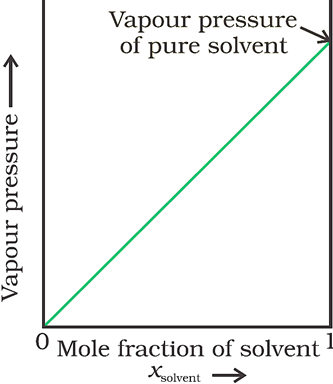
· Solutions which obey Raoult’s law over the entire range of concentrations
· For ideal solution:
· Enthalpy of mixing of the pure components to form the solution,
Δmix H = 0
· Volume of mixing, Δmix V = 0
· An ideal solution will be formed when intermolecular forces of attraction between the molecules of solute (A − A) and those between the molecules of solvent (B − B) are nearly equal to those between solute and solvent molecules (A − B).
· Examples: n-Hexane and n-heptane, bromoethane and chloroethane, benzene and toluene
· Solutions which do not obey Raoult’s law over the entire range of concentration
· The vapour pressure of a non-ideal solution is either higher or lower than that predicted by Raoult’s law.
Positive deviation from Raoult’s law
(When vapour pressure of solution is higher)
· The plot of vapour pressure of two component solutions as a function of mole fraction is shown below.
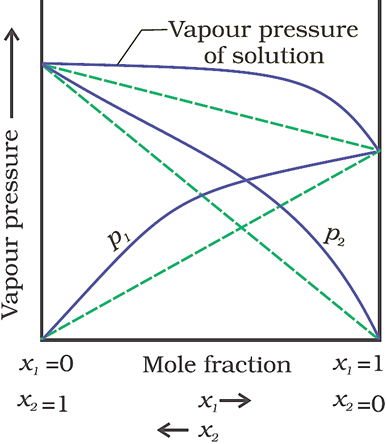
· The intermolecular forces of attraction between solute-solvent molecules are weaker than those between solute-solute molecules and solvent-solvent molecules. Therefore, solvent molecules can easily escape, resulting in increase in vapour pressure.
· Example: Ethanol and acetone mixture.
Negative deviation from Raoult’s law
(When vapour pressure of solution is lower)
· The plot of vapour pressure of two component solutions as a function of mole fraction is shown below.
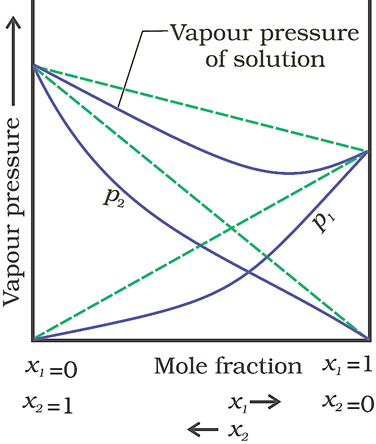
· The intermolecular forces of attraction between solute-solute molecules and solvent-solvent molecules are weaker than those between solute-solvent molecules. This results in the decreasing of vapour pressure.
· Example − Chloroform and acetone mixture
The intermolecular attractive forces between solute-solvent molecules increase due to the formation of H-bond.
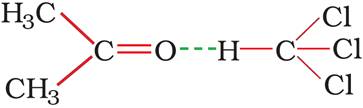
Azeotropes
· Binary mixtures which have the same composition in liquid and vapour phase, and have constant boiling points
· Not possible to separate the components by fractional distillation
· Two types – (i) Minimum boiling azeotrope (ii) Maximum boiling azeotrope
· Minimum boiling azeotrope − Solution showing a large positive deviation from Raoult’s law of specific composition
· Example: Ethanol-water mixture containing ethanol approximately 95% by volume
· Maximum boiling azeotrope − Solution showing a large negative deviation from Raoult’s law at specific composition
· Example: Nitric acid-water mixture containing 68% nitric acid and 32% water by mass
The boiling point of this azeotrope is 393.5 K.
Colligative Properties and Determination of Molar mass
· Properties which depend upon the number of solute particles present in the solution irrespective of their nature, relative to the total number of particles present in the solution
· Examples: Relative lowering of vapour pressure of the solvent, depression of freezing point of the solvent, elevation of boiling point of the solvent, osmotic pressure of the solution.
Relative Lowering of Vapour Pressure
· Relative lowering of vapour pressure is equal to the mole fraction of the solute.
i.e., 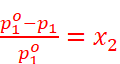
Where,
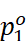 =
Vapour pressure of the solvent
=
Vapour pressure of the solvent
p1 = Vapour pressure of the solution
x2 = Mole fraction of the solute
However, 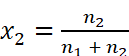
Where, n2 = Number of moles of solute
n1 = Number of moles of solvent
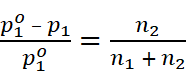
For dilute solution, n1 >> n2 , therefore
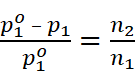
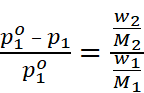
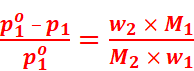
Where, w2 = Mass of solute
M2 = Molar mass of solute
w1 = Mass of solvent
M1 = Molar mass of solvent
· With addition of non-volatile solute, vapour pressure decreases and hence, boiling point increases.
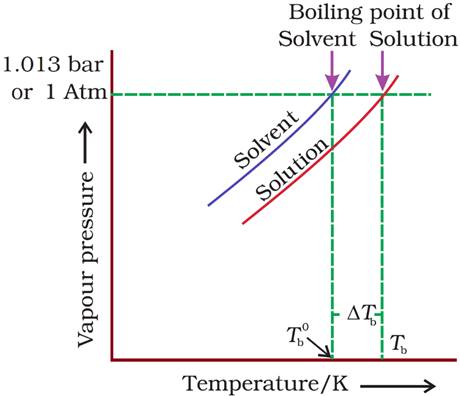
·
Here, 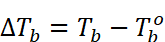 =
Elevation of boiling point
=
Elevation of boiling point
Where, Tb = Boiling of solution. Tb° = Boiling point of solvent
ΔTb ∝ m (for dilute solution)
⇒ ΔTb = Kb m
Where, m = Molality
Kb = Proportionality constant known as Boiling Point Elevation Constant
or Molal Elevation Constant or Ebullioscopic constant
(Unit = K kg mol−1)
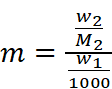
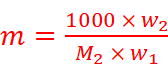
Where,
w2 = Mass of solute
M2 = Molar mass of solute
w1 = Mass of solvent
Hence,
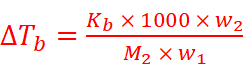
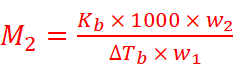
With addition of non-volatile solute, vapour pressure decreases, which leads to a decrease in freezing point.
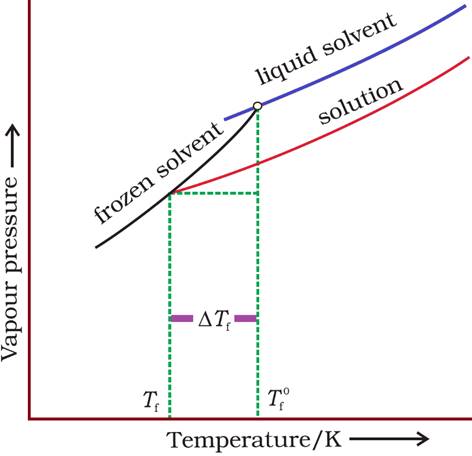
Here, 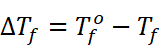 =
Depression of freezing point
=
Depression of freezing point
Where, 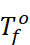 =
Freezing point of solvent
=
Freezing point of solvent
Tf = Freezing point of solution
For dilute
solution (ideal solution),
ΔTf ∝ m
⇒ ΔTf = Kf m
Where, m = Molality
Kf = Proportionality constant known as Freezing Point Constant
or Molal Depression Constant or Cryoscopic constant
(Unit = K kg mol−1)
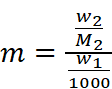
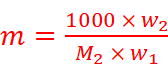
Where,
w2 = Mass of solute
M2 = Molar mass of solute
w1 = Mass of solvent
Hence,
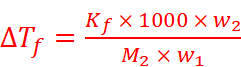
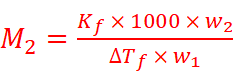
The value of Kb and Kf can be determined by the following relations:
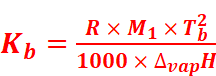
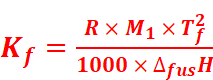
Where,
M1 = Molar mass of the solvent
R = Gas constant
ΔvapH = Enthalpy of vaporisation
ΔfusH = Enthalpy of fusion
Antifreeze solutions are added to a liquid in a cooling system (such as the water in an automobile engine) to lower its freezing point and prevent ice build-up in the system at cooler temperatures.
· It contains chemicals that prevent corrosion and scale formation in the engine and radiator of a vehicle.
· It provides protection against boiling during summers. At 1 atmosphere pressure, water boils at 100 °C; but a 50-50 blend of water−ethylene glycol boils at 106 °C.
· A mixture of antifreeze and distilled water (in the ratio, one part antifreeze to one part water) provides freeze protection down to −36.67 °C and boils over protection up to 129.4 °C.
· Avoid using concentrated antifreeze in a cooling system. At least 40% of the mixture should be water.
· Do not increase the concentration of antifreeze above 60% as it damages the freezing and overheating protection of the engine.
· Do not add too much water to the cooling system as it lowers the concentration of the corrosion inhibitor and antifreeze. This results in decreased protection against corrosion and freezing.
· The process of flow of solvent molecules from pure solvent to solution or from solution of lower concentration to solution of higher concentration through a semi-permeable membrane is called osmosis.
· The pressure required to just stop the flow of solvent due to osmosis is called osmotic pressure (π) of the solution.
· Osmotic pressure π has to be applied to the solution to prevent osmosis.
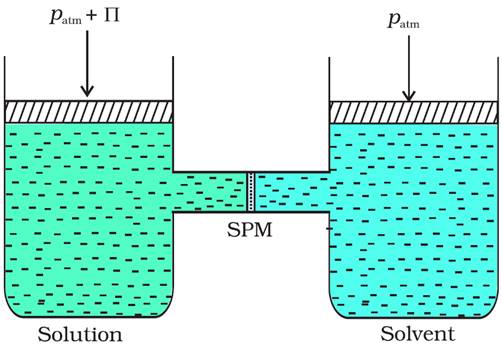
· For dilute solution, osmotic pressure is directly proportional to the molarity C of the solution at a given temperature T.
That is, π = CRT (R = Gas constant)
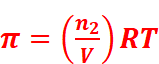
Where,
n2 = Number of moles of solute
V = Volume of the solution in litres
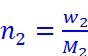
Where, w2 = Mass of the solute
M2 = Molar mass of the solute
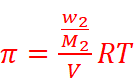
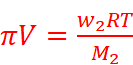
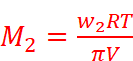
· Isotonic solutions − Solutions having same osmotic pressure. These solutions have same concentration of solute. Osmosis does not occur between two isotonic solutions.
· Hypotonic solution − Solution having a low concentration of solute relative to another.
· Hypertonic solution − Solution having a high concentration of solute relative to another.
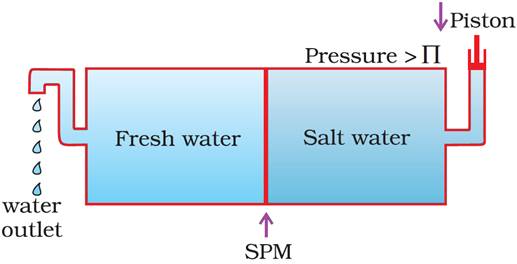
Reverse Osmosis and Water purification
· If the pressure applied on the solution is greater than its osmotic pressure, then the direction of osmosis is reversed i.e., the solvent starts passing from solution to solvent. The phenomenon is called reverse osmosis.
· This phenomenon is used in purification (desalination) of sea water.
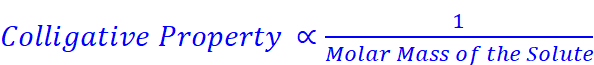
· Due to association or dissociation of molecules, the molar mass of a substance calculated from its colligative property is either lower or higher than the expected or normal value. Such molar mass is called abnormal molar mass.
Dissociation:
KCl  K+ + Cl−
K+ + Cl−
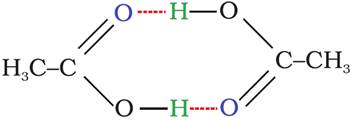
Association:
2CH3COOH ⇌ (CH3COOH)2
· To account for the extent of dissociation or association, van’t Hoff introduced a factor i, known as the van’t Hoff factor.
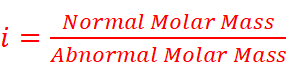
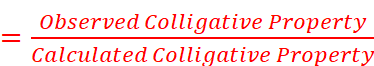
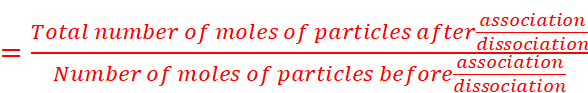
· Value of i:
For association, i < 1
For dissociation, i > 1
No association or dissociation, i = 1
· Modified equations for colligative properties after inclusion of van’t Hoff factor
· Relative lowering of vapour pressure of solvent,
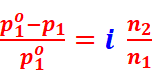
· Elevation of boiling point, ΔTb = iKb m
· Depression of freezing point, ΔTf = iKf m
·
Osmotic pressure of solution, 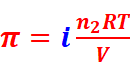
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
