Chapter 10 – Haloalkanes and Haloarenes
Important Questions – Part 5
Q.1. Write chemical equations when chlorobenzene is treated with CH3COCl in presence of anhydrous AlCl3.
Q.2. (i) Which alkyl halide from the following pairs would you expect to react more rapidly by an SN2 mechanism and why?
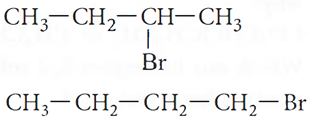
(ii) Racemisation occurs in SN1 reactions. Why?
Q.3. Write chemical equations when
(i) methyl chloride is treated with AgNO2.
(ii) bromobenzene is treated with CH3Cl in the presence of anhydrous AlCl3.
Q.4. What are ambident nucleophiles? Explain with an example.
Q.5. Chlorobenzene is extremely less reactive towards a nucleophilic substitution reaction. Give two reasons for the same.
Q.6. Haloalkanes undergo nucleophilic substitution whereas haloarenes undergo electrophilic substitution. Explain.
Q.7. Answer the following :
(i) What is known as a racemic mixture? Give an example.
(ii) Of the two bromo derivatives, C6H5CH(CH3)Br and C6H5CH(C6H5)Br, which one is more reactive in SN1 substitution reaction and why?
Q.8. Write the mechanism of the following reaction:
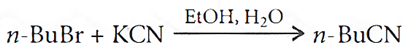
Q.9. How are the following conversions carried out?
(i) Benzyl chloride to benzyl alcohol
(ii) Methyl magnesium bromide to methylpropan-2-ol.
Q.10. Which compound in the following couple will react faster in SN2 displacement and why?
(i) 1-Bromopentane or 2-bromopentane
(ii) 1-Bromo-2-methylbutane or 2-bromo-2-methylbutane.
Q.11. Why is sulphuric acid not used during the reaction of alcohols with KI in the conversion of an alcohol to the alkyl iodide?
Q.12. Suggest a possible reason for the following observations :
(i) The order of reactivity of haloalkanes is
RI > RCl > RBr.
(ii) Neopentyl chloride (CH3)3CCH2Cl does not follow SN2 mechanism.
Q.13. Give reasons for the following observation:
The treatment of alkyl chloride with aqueous KOH leads to the formation of alcohol but in the presence of alcoholic KOH, alkene is the major product.
Q.14. (a) Identify the chiral molecule:
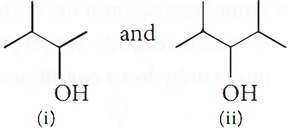
(b) Write the structure of the product when chlorobenzene is treated with methyl chloride in the presence of sodium metal and dry ether.
(c) Write the structure of the alkene formed by dehydrohalogenation of 1-bromo-1-methylcyclohexane with alcoholic KOH.
Q.15. State one use each of DDT
Q.16. State one use each of iodoform.
Q.17. Following compounds are given to you:
2-Bromopentane, 2-Bromo-2-methylbutane, 1-Bromopentane
(i) Write the compound which is most reactive towards SN2 reaction.
(ii) Write the compound which is optically active.
(iii) Write the compound which is most reactive towards β-elimination reaction.
Q.18. What is meant by chirality of a compound? Give an example.
Q.19. Rearrange the compounds of each of the following sets in order of reactivity towards SN2 displacement:
(i) 2-Bromo-2-methylbutane, 1-Bromopentane, 2-Bromopentane
(ii) 1- Bromo-3-methylbutane, 2-Bromo-2-methylbutane,3-bromo-2-methylbutane
(iii) 1-Bromobutane, 1-Bromo-2, 2-dimethylpropane, 1-Bromo-2-methylbutane
Q.20. (a) Write a chemical test to distinguish between:
(i) Chlorobenzene and benzyl chloride
(ii) Chloroform and carbon tetrachloride
(b) Why is methyl chloride hydrolysed more easily than chlorobenzene?
Q.21. Differentiate between SN1 and SN2 mechanisms and give examples.
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
