Q.1. Explain why Grignard's reagents should be prepared under anhydrous conditions.
Q.2. Predict the order of reactivity of four isomeric bromobutanes in SN1 reaction.
Q.3. Which would undergo SN2 reaction faster in the following pair and why?
CH3−CH2−Br and CH3−CH2−I
Q.4. Write the structure of an isomer of compound C4H9Br which is most reactive towards SN1 reaction.
Q.5. Out of chlorobenzene and benzyl chloride, which one gets easily hydrolysed by aqueous NaOH and why?
Q.6.
Out of 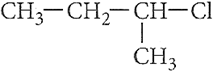 and
and 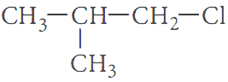 which is more reactive towards SN1 reaction and why?
which is more reactive towards SN1 reaction and why?
Q.7. Which would undergo SN2 reaction faster in the following pair and why?
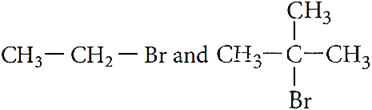
Q.8. Which would undergo SN1 reaction faster in the following pair and why?
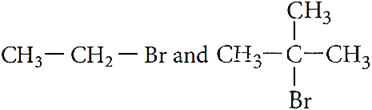
Q.9. Identify the chiral molecule in the following pair:
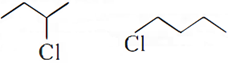
Q.10. Which halogen compound in each of the following pairs will react faster in SN2 reaction:
(i) CH3Br or CH3I
(ii) (CH3)3CCl or CH3Cl
Q.11. What happens when CH3−Br is treated with KCN?
Q.12. What happens when ethyl chloride is treated with aqueous KOH?
Q.13. Why is (±)-butan-2-ol is optically inactive?
Q.14. Which compound in the following pair undergoes faster SN1 reaction?
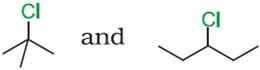
Q.15. How may methyl bromide be preferentially converted to methyl isocyanide?
Answers
Ans.1. Grignard reagents react with water to form alkanes.
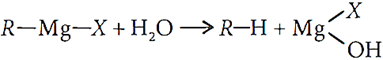
So, they must be prepared under anhydrous conditions.
Ans.2.
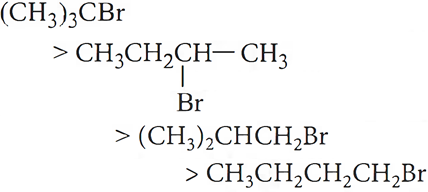
Ans.3. Since I− is a better leaving group than Br−, thus, CH3CH2I undergoes SN2 reaction faster than CH3CH2Br.
Ans.4.
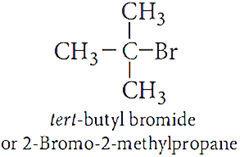
Ans.5. Benzyl chloride gets easily hydrolysed by aq. NaOH due to formation of stable benzyl carbocation. But due to partial double bond character of C−Cl bond in chlorobenzene, it does not hydrolyse.
Ans.6. The SN1 reaction proceeds through carbocation formation thus, the compound which forms more stable carbocation will be more reactive.
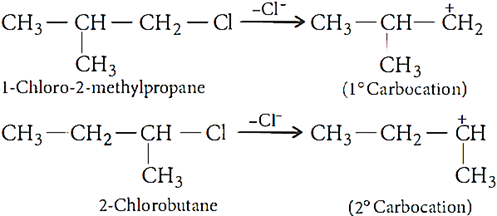
As, 2° carbocation is more stable than l° carbocation thus, 2-chlorobutane is more reactive towards SN1 reaction.
Ans.7. CH3−CH2−Br would undergo SN2 reaction faster due to formation of less sterically hindered transition state.
Ans.8.
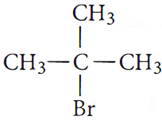 will undergo SN1
reaction faster due to the formation of stable carbocation.
will undergo SN1
reaction faster due to the formation of stable carbocation.
Ans.9.
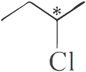 is a chiral molecule.
is a chiral molecule.
Ans.10. (i) CH3I will give faster SN2 reaction.
(ii) CH3Cl will give faster SN2 reaction.
Ans.11. CH3CN is formed by nucleophilic substitution reaction.
CH3Br + KCN → CH3CN + KBr
Ans.12. When ethyl chloride is treated with aqueous KOH, ethanol is formed,
CH3CH2Cl + KOH(aq) → CH3CH2OH + KCl
Ans.13. (±)-Butan-2-ol is optically inactive because it exists in two enantiomeric forms which are non-superimposable mirror images of each other. Both the isomers are present in equal amounts therefore, it does not rotate the plane of polarized light and is optically inactive.
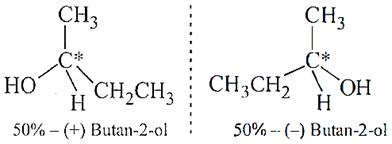
Ans.14.
Tertiary halide 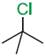 reacts faster than
the secondary halide because of the greater stability of tert-carbocation.
reacts faster than
the secondary halide because of the greater stability of tert-carbocation.
Ans.15. AgCN is mainly covalent in nature and nitrogen is free to donate electron pair forming isocyanide as main product.
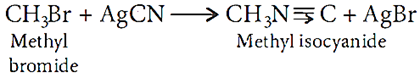
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
