Class 11 Chemistry
Final Series Assignment 15.6B
Q.1. What is the lowest value of n that allows g orbitals to exist?
Q.2. Calculate the pH of the solutions having 2 g of TlOH dissolved in water to give 2 L of solution.
Q.3. Oxidation−reduction are complementary processes. Explain.
Q.4. Write a short note on Resonance effect.
Q.5. How will you convert: ethyne to but−2−yne?
Q.6. How would you convert benzene to Hexachlorobenzene ?
Q.7. An electron is in one of the 3d orbitals. Give the possible values of n, l and ml for this electron.
Q.8. How will you convert Acetylene to chlorobenzene?
Q.9. The combination of atoms to form molecules is based on octet rule. Give two limitations of this rule.
Q.10. The following concentrations were obtained for the formation of NH3 from N2 and H2 at equilibrium at 500 K [N2] = 1.5 × 10−2 M, [H2] = 3.0 × 10−2 M and [NH3] = 1.2 × 10−2 M. Calculate equilibrium constant mention the reaction.
N2(g) + 3H2(g) ⇌ 2NH3(g)
Q.11. (ii) Draw the geometrical isomers of 2, 3−dichlorobut−2−ene
Q.12. Ka1, Ka2 and Ka3 are the respective ionization constants for the following reactions
H2S ⇌ H+ + HS−
HS− ⇌ H+ + S2−
H2S ⇌ 2H+ + S2−
Write the correct relationship between Ka1, Ka2 and Ka3.
Q.13. Why does SO3 act as an electrophile?
Q.14. Use molecular orbital theory to explain why Be2 molecule does not exist.
Q.15. On a ship sailing in Pacific Ocean where temperature is 23.4°C, a balloon is filled with 2 L air. What will be the volume of the balloon when ship reaches Indian ocean, where temperature is 26.1°C?
Q.16. Enthalpy diagram for a particular reaction is given in the figure. Is it possible to decide spontaneity of a reaction from given diagram? Explain.
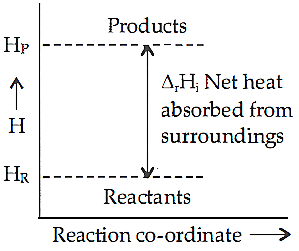
Q.17. What characteristics do you expect from an electron−deficient hydride with respect to its structure and chemical reactions ?
Q.18. The reaction 2C + O2 → 2CO is carried out by taking 24.0 g of carbon and 96.0 g of O2. Find out
(i) Which reactant is left in excess?
(ii) How much of it is left?
(iii) How many grams of the other reactant should be taken so that nothing is left at the end of the reaction?
Q.19. Predict the formulas of the stable binary compounds that would be formed by the combination of the following pairs of elements:
(a) Lithium and oxygen
(b) Magnesium and nitrogen
(c) Aluminium and iodine
(d) Silicon and oxygen
(e) Phosphorus and fluorine
(f) Element 71 and fluorine
Q.20. Predict the sign of ,entropy change in each of the following:
(i) Temperature of perfectly crystalline solid is raised from 0 K to 115 K.
(ii) 2NaHCO3(s) → Na2CO3(s) + H2O(g)
(iii) H2(g) → 2H(g)
Q.21. In three moles of ethane (C2H6), calculate the following:
(i) Number of moles of carbon atoms
(ii) Number of moles of hydrogen atoms
(iii) Number of molecules of ethane
Q.22. Element Gr 17, Gr 18, Gr 1 are in the sequence as placed in modern periodic table.
(a) Which has negative electron gain enthalpy?
(b) Which has more metallic behaviour?
(c) Which has zero electronic behaviour?
Q.23. Using s, p, d notations, describe the orbital with the following quantum numbers:
(a) n = 3, l = 1
(b) n = 4, l = 3
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE /
