Thermodynamics
Practice Question Answer Set 2
Q.1. Calculate the entropy change in surrounding when 1.00 mol of H2O(l) is formed under standard conditions. ∆fH° = – 286 kJ mol–1.
∆fH°[H2O(l)] = –286 kJ/mol
∴ qsurroundings = +286 kJ/mol
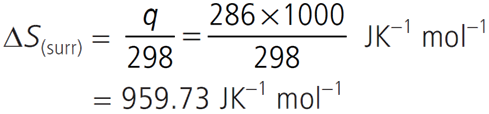
Q.2. Justify the following statements:
Many thermodynamically feasible reactions do not occur under ordinary conditions.
Under ordinary conditions, the average energy of the reactants may be less than the threshold energy. They require some activation energy to initiate the reaction.
Q.3. Justify the following statements:
At low temperature, enthalpy change dominates the value of ∆G and at high temperature it is the entropy which dominates the value of ∆G.
∆G = ∆H – T∆S
At lower temperature, if value of ∆H is negative ∆G will be –ve and if ∆H is positive, ∆G will be positive. While at higher temperature –T∆S will be high, thus, sign of ∆S will decide whether ∆G will be positive or negative.
Q.4. What is a thermochemical equation?
The balanced chemical equation which includes the amount of heat evolved or absorbed during the reaction is called a thermochemical equation.
Q.5. Write one application of Hess’s law.
Hess’s law is found very useful in calculating the enthalpy change for the reaction for which experimental determination is not possible.
Q.6. In a process, 701 J of heat is absorbed by a system and 394 J of work is done by the system. What is the change in internal energy for the process?
Heat absorbed by the system (q) = 701 J
Work done by the system (w) = –394 J
According to first law of thermodynamics,
∆U = q + w
= 701 + (–394)
= 701 – 394
= 307 J
Q.7. Derive the relationship, ∆H = ∆U + ∆ngRT.
The enthalpy H can be written as :
H = U + pV ………… (i)
For finite changes at constant pressure, we can write equation (i) as
∆H = ∆U + ∆(pV)
Since p is constant, we can write
∆H = ∆U + p∆V …………(ii)
Let us consider a reaction involving gases. If VA is the total volume of the gaseous reactants, VB is the total volume of the gaseous products, nA is the number of moles of gaseous reactants and nB is the number of moles of gaseous products, all at constant pressure and temperature, then using the ideal gas law, we write,
pVA = nART
and pVB = nBRT
Thus, pVB – pVA = nBRT – nART = (nB – nA)RT
or p(VB – VA) = (nB – nA)RT
or p∆V = ∆ngRT …………(iii)
where, ∆ng = ngaseous products – ngaseous reactants
Substituting the value of p∆V from equation (iii) in equation (ii), we get
∆H = ∆U + ∆ngRT
Q.8. 18.0 g of water completely vapourises at 100°C and 1 bar pressure and the enthalpy change in the process is 40.79 kJ mol–1. What will be the enthalpy change for vapourising two moles of water under the same conditions? What is the standard enthalpy of vaporisation for water?
Enthalpy of a reaction is the energy change per mole for the process.
18 g of H2O = 1 mole (∆Hvap = 40.79 kJ mol–1)
For 2 moles of H2O, enthalpy of vaporisation = 2 × 40.79
= 81.58 kJ
∆H°vap = 40.79 kJ mol–1
Q.9. A sample of 1.0 mol of a monoatomic ideal gas is taken through a cyclic process of expansion and compression as shown in figure. What will be the value of ∆H for the cycle as a whole?
For a cyclic process ∆H = 0
Q.10. If the combustion of 1 g of graphite produces 20.7 kJ of heat, what will be molar enthalpy change? Give the significance of sign also.
Molar enthalpy change for graphite (∆H)
= enthalpy change for 1 g × molar mass of C
= – 20.7 × 12
= – 2.48 × 102 kJ mol–1
Since the sign of ∆H = –ve, it is an exothermic reaction.
Q.11. The enthalpy of reaction for the reaction:
2H2(g) + O2(g) → 2H2O(l) is ∆rH° = –572 kJ mol–1.
What will be standard enthalpy of formation of H2O(l)?
2H2 + O2 → 2H2O; ∆rH° = 572 kJ mol–1
∆fH° will be half of the enthalpy of the given equation as enthalpy of formation is the enthalpy change of the reaction when 1 mole of the compound is formed from its elements.
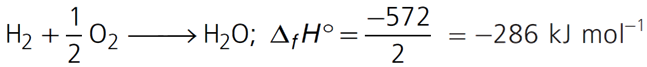
Q.12. 10 g of argon gas is compressed isothermally and reversibly at a temperature of 27 °C from 10 litre to 5 litre. Calculates q, w and ∆U for this process.
(R = 2.0 cal K–1 mol–1, atomic weight of Argon = 40)
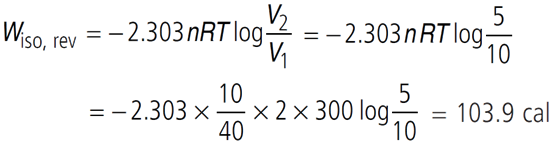
For isothermal process ∆U = 0
From 1st law of thermodynamics, ∆U = q + W
∴ q = –W = –103.9 cal
