Class 11 Chemistry
Term 2 (Assignment 2)
Q.1. The tendency of BF3 ,BCl3 and BBr3 to behave as Lewis acid decreases in the sequence
(a) BF3 > BCl3 > BBr3
(b) BCl3 > BF3 > BBr3
(c) BBr3 > BCl3 > BF3
(d) BBr3 > BF3 > BCl3
Q.2. Which of the following statements is correct?
(a) BCl3 is more stable than TlCl3
(b) AlF3 is high melting solid but AlCl3 is low melting
(c) B−X bonds distance in BX3 is shorter than the theoretical value
(d) All of the above statements
Q.3. The correct order of atomic radii in group 13 elements is
(a) B < Ga < Al < Tl < In
(b) B < Al < Ga < In < Tl
(c) B < Al < In < Ga < Tl
(d) B < Ga < Al < In < Tl
Q.4. Which of the following is a least stable compound?
(a) BCl3
(b) GaCl3
(c) InCl3
(d) TlCl3
Q.5. Which species does not exist?
(a) [BF6]3−
(b) [AlF6]3−
(c) [GaF6]3−
(d) [InF6]3−
Q.6. Sodium fire in the laboratory should not be extinguished by pouring water. Why?
Q.7. Comment on each of the following observations.
(a) The mobilities of the alkali metal ions in aqueous solution are
Li+ < Na+ < K+ < Rb+ < Cs+.
(b) Lithium is the only alkali metal which forms nitride directly.
Q.8. For an ideal gas, how does the plot of log p vs log V differ from that of log V vs log T? Explain.
Q.9. What happens, when
(a) ethyl alcohol is heated in the presence of H2SO4 at 443 K?
(b) ethyl bromide is heated with alcoholic KOH?
Q.10. Explain, why Boyle’s law cannot be used to calculate the volume of a real gas when it is converted from initial state to final state by an adiabatic expansion ?
Q.11. Predict the major product(s) of the following reactions and explain their formation.

Q.12. How would you distinguish between
(a) Be(OH)2 and Ba(OH)2
(b) BeSO4 and BaSO4
Q.13. Why does the solubility of alkaline earth metal hydroxides in water increase down the group?
Q.14. All compounds of alkali metals are easily soluble in water but lithium compounds are more soluble in organic solvents. Explain.
Q.15. Why are potassium and caesium, rather than lithium used in photoelectric cells?
Q.16. 135 mL of a gas is collected over water at 25°C and 0.993 bar. If the gas weighs 0.160 g and the aqueous tension at 25°C is 0.0317, calculate the molar mass of the gas. [Ans. 30.49 g mol−1]
Q.17. An equilibrium mixture at 300 K contains N2O4 and NO2 at 0.28 and 1.1 atm pressure respectively. If the volume of the container is doubled, calculate the new equilibrium pressure of two gases.
[Ans. p(N2O4) = 0.95 atm, p(NO2) = 0.64]
Q.18. Arrange the following in order of increasing ease of dehydrohalogenation C2H5I, C2H5Br, C2H5Cl.
Q.19. Write the structure and IUPAC name of X ?
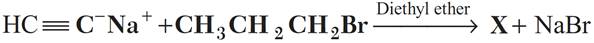
Q.20. What happens when iso-butene is treated with hot alkaline KMnO4?
Q.21. The equilibrium constant for the following reaction is 1.6 × 105 at 1024 K.
H2(g) + Br2(g) ⇌ 2HBr(g)
Find the equilibrium pressure of all gases if 10.0 bar of HBr is introduced into a sealed container at 1024 K.
[Ans. pHBr = 10 Bar]
Q.22. Explain the following
(a) Ice melts slowly at higher altitudes.
(b) Pure liquids and solids can be ignored while writing the equilibrium constant expressed.
Q.23. Bromine monochloride, BrCl decomposes into bromine and chlorine and reaches the equilibrium,
2BrCl(g) ⇌ Br2(g) + Cl2(g)
for which KC = 32 at 500 K. If initially pure BrCl is present at a concentration of 3.3 × 10−3 molL−1 , what is its molar concentration in the mixture at equilibrium? [Ans. BrCl = 2.68 ×10−4 mol L−1]
Q.24. The value of KC for the reaction.
2A ⇌ B + C is 2 × 10−3.
At a given time, the composition of reaction mixture is [A] = [B] = [C] = 3 ×10−4 M. In which direction the reaction well proceed?
Q.25. 7-bromo-1, 3, 5-cycloheptatriene exists as an ion whereas 5-bromo-1, 3-cyclopentadiene does not form an ion even in presence of Ag+. Explain.
Q.26. Explain why the following systems are not aromatic?
Q.27. An alkyne (X) has molecular formula C5H8. It reacts neither with sodamide nor with ammoniacal cuprous chloride. Write structure of X.
Q.28. A hydrocarbon ‘Y’ decolourises bromine water. On ozonolysis it gives 3-methyl butanal and formaldehyde. Give the name of the compound.
Q.29. A hydrocarbon (Z) has the molecular formula C8H10. It does not decolourise bromine water and is oxidised to benzoic acid on heating with K2Cr2O7. It can also have three other isomers A, B and C. Write the structures of Z, A, B and C.
Q.30. Which one of the following is not extensive state function?
Enthalpy change, internal energy change and pressure
Q.31. 10 moles of an ideal gas expand isothermally and reversibly from a pressure of 5 atm to 1 atm at 300 K. What is the largest mass that can be lifted through a height of 1 m by this expansion? [Ans. 4092.70 kg]
Q.32. Calculate the standard enthalpy of formation of C2H4(g) from the following thermochemical equation.

Given that ∆fH° of CO2 (g), H2O(g) as −393.5 and −249 kJ mol−1 respectively.
[Ans. ∆fH° (C2H4) =76 kJ mol−1]
Q.33. When an ideal gas expands into vacuum, there is neither absorption nor evolution of heat. Why?
Q.34. The standard heat of formation of CH4(g), CO2(g) and H2O(g) are −76.2, −394.8 and −241.6 kJ mol−1 respectively.
Calculate the amount of heat evolved by burning 1 m3 of methane measured at STP. [Ans. 35794.6 kJ]
Q.35. Predict in which of the following, entropy increases/decreases. Give reason.
(a) Temperature of crystalline solid is raised from 0 K to 115 K.
(b) H2(g) → 2H(g)
