Q.1. The equilibrium constant expression for a gas reaction is,
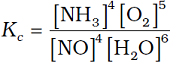
Write the balanced chemical equation corresponding to this expression.
Q.2. One mole of H2O and one mole of CO are taken in 10 L vessel and heated to 725 K. At equilibrium 40% of water (by mass) reacts with CO according to the equation,
H2O(g) + CO(g) ⇌ H2(g) + CO2(g)
Calculate the equilibrium constant for the reaction.
Q.3. At 700 K, equilibrium constant for the reaction:
H2(g) + I2(g) ⇌ 2HI(g)
is 54.8. If 0.5 mol L –1 of HI(g) is present at equilibrium at 700 K, what are the concentration of H2(g) and I2(g) assuming that we initially started with HI(g) and allowed it to reach equilibrium at 700K?
Q.4. What is the equilibrium concentration of each of the substances in the equilibrium when the initial concentration of ICl was 0.78 M ?
2ICl(g) ⇌ I2(g) + Cl2(g); Kc = 0.14
Q.5. Kp = 0.04 atm at 899 K for the equilibrium shown below. What is the equilibrium concentration of C2H6 when it is placed in a flask at 4.0 atm pressure and allowed to come to equilibrium?
C2H6(g) ⇌ C2H4(g) + H2(g)
Q.6. Ethyl acetate is formed by the reaction between ethanol and acetic acid and the equilibrium is represented as:
CH3COOH(l) + C2H5OH(l) ⇌ CH3COOC2H5(l) + H2O(l)
(i) Write the concentration ratio (reaction quotient), Qc, for this reaction (note: water is not in excess and is not a solvent in this reaction)
(ii) At 293 K, if one starts with 1.00 mol of acetic acid and 0.18 mol of ethanol, there is 0.171 mol of ethyl acetate in the final equilibrium mixture. Calculate the equilibrium constant.
(iii) Starting with 0.5 mol of ethanol and 1.0 mol of acetic acid and maintaining it at 293 K, 0.214 mol of ethyl acetate is found after sometime. Has equilibrium been reached?
Q.7. A sample of pure PCl5 was introduced into an evacuated vessel at 473 K. After equilibrium was attained, concentration of PCl5 was found to be 0.5 × 10–1 mol L–1. If value of Kc is 8.3 × 10–3, what are the concentrations of PCl3 and Cl2 at equilibrium?
PCl5(g) ⇌ PCl3(g) + Cl2(g)
Q.8. One of the reactions, that takes place in producing steel from iron ore is the reduction of iron(II) oxide by carbon monoxide to give iron metal and CO2.
FeO(s) + CO(g) ⇌ Fe(s) + CO2(g); Kp = 0.265 atm at 1050K
What are the equilibrium partial pressures of CO and CO2 at 1050 K if the initial partial pressures are: pCO = 1.4 atm and pCO2 = 0.80 atm?
Q.9. Equilibrium constant, Kc for the reaction
N2(g) + 3H2(g) ⇌ 2NH3(g) at 500 K is 0.061
At a particular time, the analysis shows that composition of the reaction mixture is 3.0 mol L–1 N 2, 2.0 mol L–1 H2 and 0.5 mol L–1 NH 3. Is the reaction at equilibrium?
If not in which direction does the reaction tend to proceed to reach equilibrium?
Q.10. Bromine monochloride, BrCl decomposes into bromine and chlorine and reaches the equilibrium:
2BrCl(g) ⇌ Br2(g) + Cl2(g)
for which Kc = 32 at 500 K. If initially pure BrCl is present at a concentration of 3.3 × 10–3 mol L–1, what is its molar concentration in the mixture at equilibrium?
Q.11. At 1127 K and 1 atm pressure, a gaseous mixture of CO and CO2 in equilibrium with solid carbon has 90.55% CO by mass
C(s) + CO2(g) ⇌ 2CO(g)
Calculate Kc for this reaction at the above temperature.
Q.12. Calculate
a) ∆Go and b) the equilibrium constant for the formation of NO2 from NO and O2 at 298K
NO(g) + ½ O2(g) ⇌ NO2(g)
Where ∆fGo(NO2) = 52.0 kJ/mol
∆fGo(NO) = 87.0 kJ/mol
∆fGo(O2) = 0 kJ/mol
Online Tuitions and Self-Study Courses for Grade 6 to 12 & JEE / NEET
