Mendeleev made a successful effort in grouping elements in the form of rows and columns (periodic table).
Limitations of Mendeleev’s periodic table are listed below:
· The position of hydrogen was not justified in Mendeleev’s periodic table.
· The discovery of isotopes revealed another limitation of Mendeleev’s periodic table.
· Although Mendeleev arranged the elements in the increasing order of their atomic masses, there were instances where he had placed an element with a slightly higher atomic mass before an element with a slightly lower atomic mass.
In 1913, Henry Moseley demonstrated that atomic number (instead of atomic mass) is a more fundamental property for classifying elements.
The atomic number of an element is equal to the number of protons present in an atom of that element. Since the number of protons and electrons in an atom of an element is equal, the atomic number of an element is equal to the number of electrons present in a neutral atom.
Atomic number = Number of protons = Number of electrons
The number of protons or electrons in an element is fixed. No two elements can have the same atomic number. Hence, elements can be easily classified in the increasing order of their atomic numbers.
The modern periodic law states that
The properties of elements are a periodic function of their atomic numbers.

All elements in the same group contain the same number of valence electrons.
E.g., group two elements beryllium, magnesium, calcium, etc. contain the same number of valence electrons. The number of valence electrons present in these elements is 2.
On the other hand, the number of shells increases as we go down the group.
All elements in the same period contain the same valence shell.
E.g., in the third period of the modern periodic table, elements sodium, magnesium, aluminium, silicon, phosphorus, sulphur, chlorine, and argon contain the same number of valence shells, but they do not have the same number of valence electrons.
|
Name of the element |
Electronic configuration (K, L, M) |
|
Sodium |
2, 8, 1 |
|
Magnesium |
2, 8, 2 |
|
Aluminium |
2, 8, 3 |
|
Silicon |
2, 8, 4 |
|
Phosphorus |
2, 8, 5 |
|
Sulphur |
2, 8, 6 |
|
Chlorine |
2, 8, 7 |
|
Argon |
2, 8, 8 |
Anomalous behaviour of hydrogen
In the modern periodic table, hydrogen is placed above alkali metals because of resemblance with their electronic configurations. However, it is never regarded as an alkali metal. This makes hydrogen a unique element.
In some modern periodic tables hydrogen is placed above rest of the elements as it shows similarities and dissimilarities with both Group 1 (alkali metals) and Group 17 (Halogens).
Calculation of number of elements present in the first, second, third, and fourth periods:
The number of electrons that a shell can hold can be calculated using the formula 2n2. Here, n represents the number of shells from the nucleus. For example, n is equal to 1, 2, and 3 for K, L, and M shells respectively. Hence, the number of electrons that each of these shells can hold can be calculated by substituting the value of n in the given formula.
Number of electrons that K shell can accommodate = 2n2 = 2 × 12 = 2
Hence, K shell can accommodate only 2 electrons and only two elements are present in the first period.
Similarly, second and third periods can accommodate 8 and 18 electrons respectively.
Since their outermost shells can contain only 8 electrons, there are only 8 elements in both the periods.
The position of an element in the Modern Periodic Table tells us about its chemical reactivity.
The valence electrons determine the kind and the number of bonds formed by an element.
IUPAC Notation for Elements with Atomic Number > 100
|
Digit |
Name |
Abbreviation |
|
0 |
nil |
n |
|
1 |
un |
u |
|
2 |
bi |
b |
|
3 |
tri |
t |
|
4 |
quad |
q |
|
5 |
pent |
p |
|
6 |
hex |
h |
|
7 |
sept |
s |
|
8 |
oct |
o |
|
9 |
enn |
e |
· Latin words for various digits of the atomic number are written together in the order of digits, which make up the atomic number, and at the end, ‘ium’ is added.
Nomenclature of Elements with Atomic Number Above 100
|
Atomic number |
Name |
Symbol |
IUPAC Official Name |
IUPAC Symbol |
|
101 |
Unnilunium |
Unu |
Mendelevium |
Md |
|
102 |
Unnilbium |
Unb |
Nobelium |
No |
|
103 |
Unniltrium |
Unt |
Lawrencium |
Lr |
|
104 |
Unnilquadium |
Unq |
Rutherfordium |
Rf |
|
105 |
Unnilpentium |
Unp |
Dubnium |
Db |
|
106 |
Unnilhexium |
Unh |
Seaborgium |
Sg |
|
107 |
Unnilseptium |
Uns |
Bohrium |
Bh |
|
108 |
Unniloctium |
Uno |
Hassnium |
Hs |
|
109 |
Unnilennium |
Une |
Meitnerium |
Mt |
|
110 |
Ununnillium |
Uun |
Darmstadtium |
Ds |
|
111 |
Unununnium |
Uuu |
Rontgenium |
Rg |
|
112 |
Ununbium |
Uub |
|
|
|
113 |
Ununtrium |
Uut |
|
|
|
114 |
Ununquadium |
Uuq |
|
|
|
115 |
Ununpentium |
Uup |
|
|
|
116 |
Ununhexium |
Uuh |
|
|
|
117 |
Ununseptium |
Uus |
|
|
|
118 |
Ununoctium |
Uuo |
|
|
Electronic Configuration in Periods
· Period indicates the value of ‘n’ (principal quantum number) for the outermost or valence shell.
· Successive period in the periodic table is associated with the filling of the next higher principal energy level (n = 1, n = 2, etc.). e.g.,
First period (n = 1) → hydrogen (1s1) and helium (1s2) [2 elements]
Second period (n = 2) → Li (1s2 2s1), Be (1s2 2s2), B (1s2 2s2 2p1) to Ne (2s2 2p6) [8 elements]
3d orbital is filled up before 4p orbitals (3d orbitals → energetically favourable)
Hence these elements are known as 3d-transition series
Sc (3d1 4s2) to Zn (3d10 4s2) (n = 3)
4d-transition series starts at Ytterbium and ends at Cadmium.
Elements from Z = 58 to Z = 71 are called 4f-inner transition series or lanthanoid series (filling up of the 4f orbitals).
Filling up of 5f orbitals after Ac (Z = 89) gives 5f-inner transition series or the actinoid series.
Electronic Configuration in Groups
· Same number of electrons is present in the outer orbitals (that is, similar valence shell electronic configuration).
· Electronic configuration of group 1 elements is given in the following table.
|
Atomic number |
Symbol |
Electronic configuration |
|
3 |
Li |
1s2 2s1(or) [He]2s1 |
|
11 |
Na |
1s2 2s2 2p6 3s1(or) [Ne]3s1 |
|
19 |
K |
1s2 2s2 2p6 3s2 3p6 4s1(or) [Ar]4s1 |
|
37 |
Rb |
1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 5s1(or) [Kr]5s1 |
|
55 |
Cs |
1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 6s1(or)[Xe]6s1 |
|
87 |
Fr |
[Rn]7s1 |
Electronic Configurations and Types of Elements
· s- Block Elements
· Group 1 (alkali metals) − ns1 (outermost electronic configuration)
· Group 2 (alkaline earth metals) − ns2 (outermost electronic configuration)
· Alkali metals form +1 ion and alkaline earth metals form +2 ion.
· Reactivity increases as we move down the group.
· They are never found in pure state in nature. (Reason − they are highly reactive)
· p - Block Elements
· Elements belonging to Groups 13 to 18
· Outermost electronic configuration varies from ns2np1 to ns2np6 in each period.
· Group 18 (ns2np6) − noble gases
· Group 17 − halogen
· Group 16 − chalcogens
· Non-metallic character increases from left to right across a period.
· d- Block Elements (Transition Elements)
· Elements of group 3 to group 12
· General electronic configuration is (n − 1) d1−10 ns0-2.
· Called transition elements
· Zn, Cd, and Hg with (n − 1) d10 ns2 configuration do not show properties of transition elements.
· All are metals. They form coloured ions, exhibit variable oxidation states, paramagnetism, and are used as catalysts.
· f- Block Elements
· Lanthanoids → Ce (Z = 58) to Lu (Z = 71)
· Actinoids → Th (Z = 90) to Lr (Z = 103)
· Outer electronic configuration → (n − 2) f1−14 (n −1) d0−1 ns2
· They are called inner-transition elements.
· All are metals.
· Actinoid elements are radioactive.
· Elements after uranium are called Transuranium elements.
Metals, Non-metals, and Metalloids
· Metals → Appear on the left side of the periodic table
· Non-metals → Located at the top right-hand side of the periodic table
· Elements change from metallic to non-metallic from left to right.
· Elements such as Si, Ge, As, Sb, Te show the characteristic properties of both metals and non-metals. They are called semi-metals or metalloids.
Periodic Trends in Physical Properties
· Atomic radii decrease with the increase in the atomic number in a period.
· For example, atomic radii decrease from Li to F in the second period.
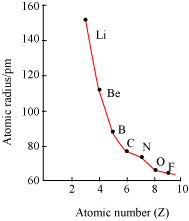
· Nuclear charge increases progressively by one unit in moving from left to right across the period. As a result, the electron cloud is pulled closer to the nucleus by the increased effective nuclear charge, which causes decrease in atomic size.
· Atomic radii increase from top to bottom within a group of the periodic table.
· Variation of atomic radii with atomic number among alkali metals and halogen:

· Cation is smaller than its parent atom.
· The size of the anion is larger than its parent atom.
· Defined as the amount of energy required to remove the most loosely bound electron from the isolated gaseous atom in its ground state
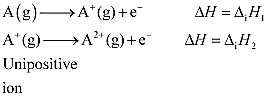
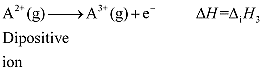
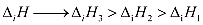
Properties of Ionization Enthalpy (I.E.):
· Decreases with the increase in atomic size
· Increases with the increase in nuclear charge
· Decreases with the increase in the number of inner electrons
· Increases with the increase in penetration power of electrons
· Atom having a more stable configuration has high value of enthalpy.
· Variation across a period: Increases with the increase in atomic number across the period.
· Variation in a group: Decreases regularly with the increase in atomic number within a group.
· Defined as the enthalpy change taking place when an isolated gaseous atom accepts an electron to form a monovalent gaseous anion
X(g) + e− → X−(g)
· Larger the value of electron gain enthalpy, greater is the tendency of an atom to accept electron.
· Greater the magnitude of nuclear charge, larger will be the negative value of electron gain enthalpy.
· Larger the size of the atom, smaller will be the negative value of electron gain enthalpy.
· More stable the electronic configuration of the atom, more positive will be the value of its electron gain enthalpy.
· Variation across a period − Tends to become more negative as we go from left to right across a period
· Variation down a group − Becomes less negative on going down the group
· Defined as the tendency of an atom in a molecule to attract the shared pair of electrons towards itself
· Greater the effective nuclear charge, greater is the electronegativity.
· Smaller the atomic radius, greater is the electronegativity.
· In a period − Increases on moving from left to right
· In a group − Decreases on moving down a group
Non −Metallic (and Metallic Character) of an Element
· Non-metallic elements have strong tendency to gain electrons.
· Non-metallic character is directly related to electronegativity and metallic character is inversely related to electronegativity.
· Across a period, electronegativity increases. Hence, non-metallic character increases (and metallic character decreases).
· Down a group, electronegativity decreases. Hence, non-metallic character decreases (and metallic character increases).
The periodic trends of various properties of elements in the periodic table are shown in figure.
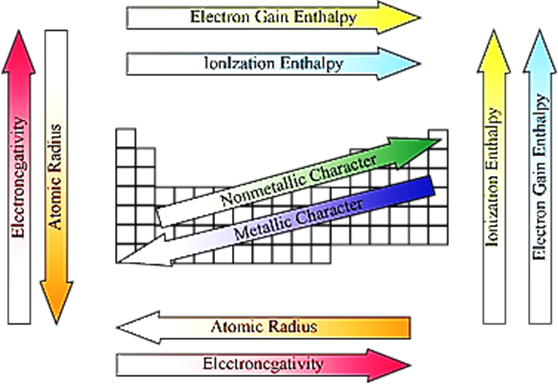
Periodic Trends in Chemical Properties
Periodicity of Valence or Oxidation States
Valence of the elements = Number of electrons in the outermost orbitals (if valence electrons ≤ 4)
Or, valency of the element = 8 − Number of outermost electrons (if valence electrons > 4)
|
Group |
1 |
2 |
13 |
14 |
15 |
16 |
17 |
18 |
|
Number of valence electrons |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
|
Valence |
1 |
2 |
3 |
4 |
3,5 |
2,6 |
1,7 |
0,8 |
Table shows the periodic trends observed in the valence of elements (hydrides and oxides).
|
Group |
1 |
2 |
13 |
14 |
15 |
16 |
17 |
|
Formula of hydride |
LiH |
|
B2H6 |
CH4 |
NH3 |
H2O |
HF |
|
|
NaH |
CaH2 |
AlH3 |
SiH4 |
PH3 |
H2S |
HCl |
|
|
KH |
|
|
GeH4 |
AsH3 |
H2Se |
HBr |
|
|
|
|
|
SnH4 |
SbH3 |
H2Te |
HI |
|
Formula of oxide |
Li2O |
MgO |
B2O3 |
CO2 |
N2O3, N2O5 |
|
|
|
|
Na2O |
CaO |
Al2O3 |
SiO2 |
P4O6, P4O10 |
SO3 |
Cl2O7 |
|
|
K2O |
SrO |
Ga2O3 |
GeO2 |
As2O3, As2O5 |
SeO3 |
|
|
|
|
BaO |
In2O3 |
SnO2 |
Sb2O3, Sb2O5 |
TeO3 |
|
|
|
|
|
|
PbO2 |
Bi2O3 |
|
|
· Many elements exhibit variable valence (particularly transition elements and actinoids).
Anomalous Properties of Second Period Elements
· First member of each group (the element in the second period from lithium to fluorine) differs in many respects from the rest of the members of the same group.
· For example, the behaviour of Li and Be is more similar with the second element of the following group i.e., Mg and Al respectively.
· Such sort of similarity is commonly known as diagonal relationship.
· Reasons for the different chemical behaviour of the first member of a group of elements in the s- and p-blocks as compared to the other members in the same group are:
· Small atomic size of the first element
· Large charge/radius ratio
· High electronegativity
· Absence of d-orbitals in the valence shell
· Ability to form pπ − pπ multiple bonds
· First member of each group of p-block element has the tendency to form pπ − pπ multiple bonds to itself and to the other second period elements. For example, C = C, C ≡ C, C = O, C = N
· Reason − This property of the elements is due to their small size.
· Higher members of the group have little tendency to form pπ − pπ bonds.
Periodic Trends and Chemical Reactivity
· High chemical reactivity at the two extremes of a period and the lowest in the centre
· Maximum chemical reactivity is at the extreme left of a period because of the ease of electron loss (or low ionization enthalpy).
· Elements at the extreme left exhibit strong reducing behaviour and elements at the extreme right exhibit strong oxidizing behaviour.
· Oxides formed by elements on left are basic and by elements on right are acidic in nature.
· Oxides of elements in the centre are amphoteric or neutral.
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
