Q.1. Why does cis-2-butene have a higher boiling point than trans-2-butene?
Ans: cis-2-Butene is more polar than trans-2-butene, therefore, it has more van der Waals' forces of attraction, hence higher boiling point.
Q.2. Why has trans-isomer higher melting point than cis-isomer?
Ans: Trans-isomer, being symmetrical, can fit into crystal lattice more readily than cis-form, therefore, it has higher melting point.
Q.3. Which of the following exhibits geometrical isomerism?
(i) 2-Methylpropene (ii) 1-Butene (iii) 2-Butene (iv) 2, 3-Dibromo-2-butene
Ans: (iii) 2-Butene and (iv) 2, 3-dibromo-2-butene can exhibit geometrical isomerism.
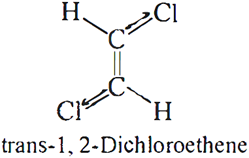
Q.4. Why is dipole moment of trans-1, 2-dichloroethene zero?
Ans: It is because individual dipoles are equal and opposite and net dipole moment is zero.
Q.5. Which type of isomerism is present in CH3CH2OH and CH3OCH3?
Ans: Functional isomerism.
Q.6. Explain why n-pentane has higher boiling point than neo-pentane.
Ans: n-Pentane has more surface area than neo-pentane, therefore, has more van der Waals' forces of attraction hence has higher boiling point.
Q.7. What is the number of σ and π-bonds in N≡C–CH=CH–C≡N?
Ans: There are 7 σ-bonds and 5 π-bonds.
Q.8. Why eclipsed and staggered forms of ethane cannot be isolated at room temperature?
Ans: It is because the difference in their energy is very small (approx. 12.5 kJ mole–1) and hence they can interconvert without any external energy.
Q.9. How many secondary carbons are there in 2, 2-dimethylbutane?
Ans: One.
Q.10. What is the hybridisation of central carbon in 1,2-propadiene (CH2=C=CH2)?
Ans: sp.
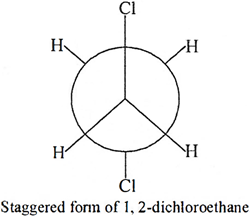
Q.11. Draw the Newman's projection formula of the staggered form of 1, 2-dichloroethane.
Ans:
Q.12. Name the type of hybridisation between C(2) and C(3) in the following molecule:
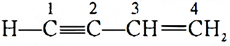
Ans: C(2) is sp hybridised and C(3) is sp2 hybridised
Q.13. Which type of isomerism is exhibited by but-1-yne and but-2-yne?
Ans: Positional isomerism is shown by but-1-yne and but-2-yne.
Q.14. Why do hydrocarbon molecules with an odd number of carbon atoms have lower melting points than those with an even number of carbon atoms?
Ans: Molecules with odd number of carbon atoms have lower melting points because they do not fit into crystal lattice easily whereas, hydrocarbons with even number of carbon atoms can fit into crystal lattice easily.
Q.15. Why is addition reaction of bromine to benzene difficult?
Ans: Addition reaction of bromine to benzene is difficult due to delocalisation of π-electrons. It does not have pure double bonds.
Q.16. Why branched chain hydrocarbons have lower boiling point than straight chain hydrocarbons?
Ans: Branched chain hydrocarbons try to acquire spherical shape which has minimum surface area, therefore, minimum van der Waals' forces of attraction, hence, lower boiling point.
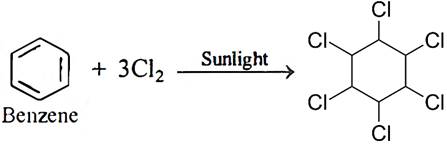
Q.17. What happens when benzene is treated with excess of Cl2 in presence of sunlight? Give chemical reaction.
Ans: BHC (Benzene hexachloride) is formed.
Q.18. What is decarboxylation? Give an example.
Ans: The process of removing carbon dioxide from sodium salt of acid with the help of soda lime is called decarboxylation.
CH3COONa
+ NaOH(CaO)  CH4 + Na2CO3
CH4 + Na2CO3
Q.19. Convert ethyne to propyne.
Ans:
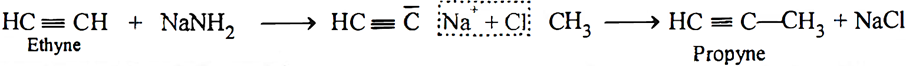
Q.20. What is Lindlar's catalyst? Give its use.
Ans: A Lindlar’s catalyst is a heterogeneous catalyst that consists of palladium deposited on barium sulfate (Pd/BaSO4) or calcium carbonate (Pd/CaCO3). It is used to convert alkynes to alkenes with the help of H2.
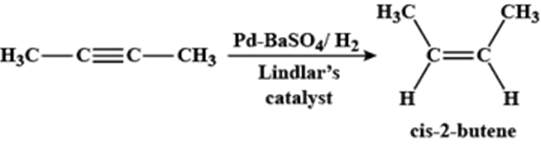
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
