Q.1.
Write IUPAC names of the following:
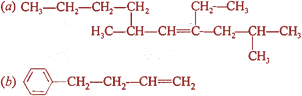
Q.2.
Give mechanism of electrophilic substitution reaction of
halogenation of arenes.
Q.3. What is hyperconjugation effect? How does it differ from resonance effect?
Q.4. How are free radicals, carbocations and carbanions produced?
Q.5. Write the IUPAC name of
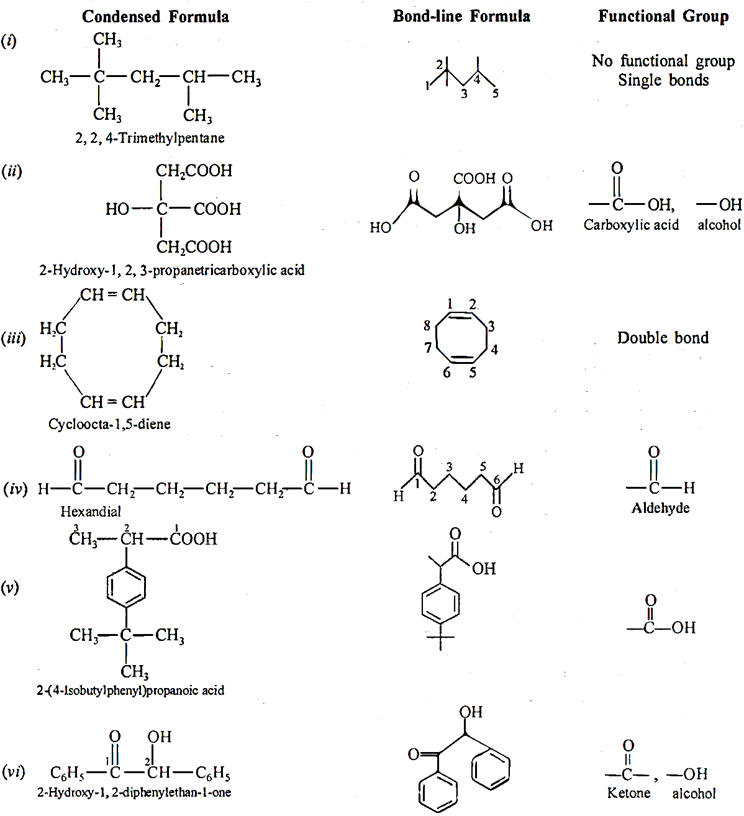
Q.6. Give condensed and bond-line structural formulae and identify the functional group(s) present, if any, for:
(i) 2, 2,4-trimethylpentane
(ii) 2-hydroxy-1, 2, 3-propanetricarboxylic acid
(iii) Cycloocta-1 ,5-diene
(iv) Hexandial
(v) 2-(4-isobutylphenyl) propanoic acid
(vi) 2-hydroxy-1, 2-diphenylethan-l-one.
Q.7. Write IUPAC name of the following compounds:
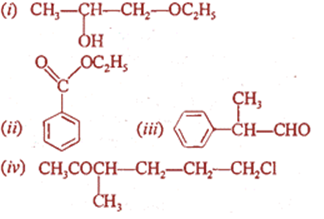
Q.8. Arrange the following in increasing order of stability:
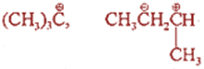 and
and Q.9.
Arrange the following in increasing acidic strength
CH3CH2COOH, (CH3)2CHCOOH and (CH3)3CCOOH
Q.10. Write the name of isomerism of the following compounds:
Q.11. What type of isomerism is shown by pentane and 2-methylbutane (isopentane)?
Q.12. Write the IUPAC name of CH2=CHCH2CH(OH)CH3.
Q.13. Write IUPAC name of the following compounds:
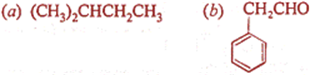
Q.14. What is functional isomerism? Give an example.
Q.15. What are electrophiles and nucleophiles? Give one example of each.
Q.16. Which of the following carbocations is most stable:
Q.17. Give one example of electrophilic substitution reaction.
Q.18. Give one example of position isomerism.
Q.19. Give one example of nucleophilic addition reaction.
Q.20. Give one example of functional isomerism.
Answers
Ans 1:
(a) 4-Ethyl-2,6-dimethyldec-4-ene
(b) 4-Phenylbut-l-ene
Ans 2:
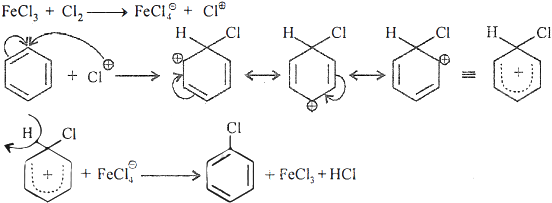
Ans 3:
Hyper conjugation Effect: It involves
orbital interaction between π electrons σ-bonds of substituent groups in
organic compound. It has no bond resonance.
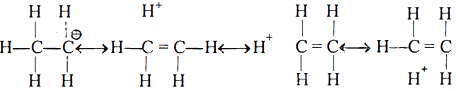
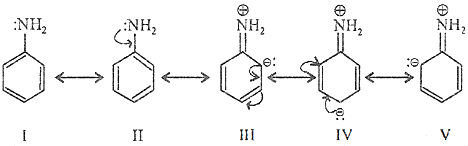
Resonance Effect: It involves π electrons and lone pair of electrons and leads to delocalisation of electrons. Resonating Structure of Aniline:
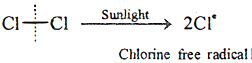
Ans 4: Free radicals are produced by homolytic fission, e.g.
Carbocations are formed by heterolytic fission, e.g.
Carbanions are formed by abstraction of H + by nucleophile from α-carbon (carbon attached to functional group).
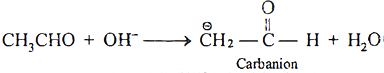
Ans 5: 3-Chloropropanal
Ans 6:
Ans 7:
(i) 1-Ethoxypropan-2-ol
(ii) Ethylbenzoate
(iii) 2-Phenylpropanal
(iv) 6-Chloro-3-methylhexan-2-one
Ans 8:
![]()
Ans 9:
![]()
Ans 10:
Metamerism.
Ans 11: Chain isomerism.
Ans 12: Pent-4-en-2-ol.
Ans 13:
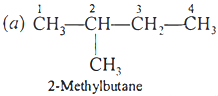 and
and 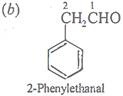
Ans 14: When two or more compounds have the same molecular formula but different
functional groups, they are said to show functional isomerism. e.g., the
molecular formula C3H6O represents an aldehyde and a
ketone.
Ans 15: A reagent that has an electron pair is called a nucleophile. A nucleophile is nucleus loving species. It is either negatively charged or electron rich species e.g., Cl–, :NH3, OH–.
A reagent that takes away an electron pair is called electrophile. An electrophile is an electron loving species. It is either positively charged or electron deficient e.g., NO2+, BF 3.
Ans 16: (CH3)3+C being a tertiary carbonium ion is most stable. The contributing factor towards maximum stability of tertiary carbonium ion is maximum number of hyperconjugative structures due to presence of large number of C – H bonds. Secondly, –CH3 group is electron releasing which stabilises intermediate carbocation.
Ans 17:
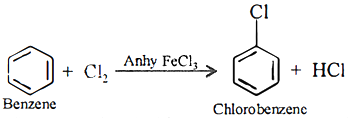
Ans 18:

Ans 19:
![]()
Ans 20:
CH3CH2OH and CH3OCH3 are
examples of functional isomers.
Online Tuitions and Self-Study Courses for Grade 6 to 12 & JEE / NEET
