Class 11 Chemistry
Chapter 11 – The p-Block Elements
Very Important Questions
Q.1. Why do boron halides form addition compounds with ammonia and amines?
Ans: It is because boron halides are electron deficient and nitrogen in ammonia and amines has 2 unpaired electrons.
Q.2. What is the hybridisation of carbon in diamond?
Ans: sp3.
Q.3. What name is given to the compounds formed by more electropositive elements with carbon?
Ans: Ionic compounds
Q.4. Why is BF3 weaker Lewis acid than BCl3?
Ans: BF3 is weaker Lewis acid than BCl3 because of more effective back bonding in case of F due to smaller size than Cl.
Q.5. Mention the chief reason for the anomalous behaviour of boron in Group 13 of the periodic table.
Ans: It is due to small size and higher ionisation energy and high charge/size ratio of boron.
Q.6. Mention the type of hybrid orbitals of silicon in SiF62– ion.
Ans: sp3d2.
Q.7. Mention the state of hybridization of B in BH4–.
Ans: sp3.
Q.8. How is that silicon atoms can have a coordination number more than four but carbon atoms cannot?
Ans: Silicon has vacant d-orbitals; therefore, it can have coordination number more than four but carbon cannot have because it does not have d-orbitals.
Q.9. Between AlF3 and AlCl3, which one will have a higher melting point?
Ans: AlF3 is more ionic, therefore, has higher melting point.
Q.10. Which one of the following elements exhibits +1 oxidation state as well?
Al, B, Ca, Tl, Be
Ans: Tl shows +l oxidation state.
Q.11. Why does boron trifluoride behave as Lewis acid?
Ans: It is because 'B' shares three electrons with fluorine and its octet is not complete, therefore, it behaves as Lewis acid.
Q.12. Why is boron metalloid?
Ans: Boron resembles both with metals and non-metals, therefore, it is metalloid.
Q.13. Name two isotopes of boron.
Ans: 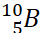 and
and 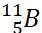 are isotopes of boron.
are isotopes of boron.
Q.14. Why does boron not form B3+ ion?
Ans: Boron does not form B3+ ion due to smallest atomic size and highest ionisation enthalpy.
Q.15. Why is crystalline boron hard solid?
Ans: It is due to strong covalent bonds.
Q.16. Why does boron resemble Si?
Ans: Both have similar charge over radius ratio, i.e., similar polarising power.
Q.17. Why does boron form stable electron deficient compounds?
Ans: Boron has three valence electrons; it will share three electrons with other elements to form electron deficient compounds which are stable.
Q.18. Why is boric acid (H3BO3) monobasic acid?
Ans: It accepts a pair of electrons from OH– ion of H2O therefore, it is monobasic acid.
B(OH)3 + H2O → [B(OH)4]– + H–
Q.19. What type of glass is obtained when borax is added to sodium carbonate, silica and calcium carbonate?
Ans: Pyrex glass is obtained which is heat resistant. It can withstand high temperature.
Q.20. Why does BF63– not exist?
Ans: It is because 'B' does not have vacant d-orbitals.
Q.21. Carbon and silicon are mainly tetravalent but Ge, Sn and Pb show divalency. Give reason.
Ans: Ge, Sn, Pb are divalent due to inert pair effect which is not there in carbon and silicon.
Q.22. What property of anhydrous AlCl3 makes it a very good preparative reagent in organic chemistry?
Ans: It acts as Lewis acid.
Q.23. Although pure silicon is an insulator, then how does it behave as semiconductor on heating?
Ans: Silicon becomes semiconductor on heating because electrons become free to move.
Q.24. What is the oxidation state of Ni in [Ni(CO)4]?
Ans: Ni has oxidation state of zero in [Ni(CO)4].
Q.25. Why does C differ from rest of elements?
Ans: Carbon has smallest size, highest ionisation enthalpy and high electronegativity and does not have d-orbitals, therefore, it differs from rest of the elements.
Q.26. Arrange the following in increasing order of Lewis acid character:
BF3, BCl3, BBr3 and BI3
Ans: BF3 < BCl3 < BBr3 < BI3.
Q.27. BCl3 exists but BH3 does not. Explain.
Ans: In BCl3, Cl donates lone pair of electrons to vacant p-orbital of boron (back-bonding) making it more stable whereas in BH3 back-bonding is not possible, therefore, it exists as dimer. Secondly, in BCl3, chlorine being bigger in size cannot form bridged bonds.
Q.28. Arrange the hydrides of group 14 in increasing order of thermal stability.
Ans: PbH4 < SnH4 < GeH4 < SiH4 < CH4.
Q.29. Arrange the hydrides of group 14 in increasing order of reducing power.
Ans: CH4 < SiH4 < GeH4 < SnH4 < PbH4.
Q.30. Why is PbCl4 good oxidizing agent?
Ans: PbCl4 is good oxidising agent because Pb2+ is more stable than Pb4+ due to inert pair effect, therefore, Pb4+ readily changes into Pb2+ by gaining two electrons.
Q.31. An aqueous solution of borax is
(a) neutral (b) amphoteric (c) basic (d) acidic
Ans: (c)
Q.32. Boric acid is polymeric due to
(a) its acidic nature (b) the presence of hydrogen bonds
(c) its monobasic nature (d) its geometry
Ans: (b)
Q.33. The type of hybridisation of boron in diborane is
(a) sp (c) sp3 (b) sp3 (d) dsp2
Ans: (c)
Q.34. Thermodynamically, the most stable form of carbon is
(a) diamond (b) graphite (c) fullerenes (d) coal
Ans: (b)
Q.35. Elements of group 14
(a) exhibit oxidation state of +4 only
(b) exhibit oxidation state of +2 and +4
(c) form M2– and M4+ ions
(d) form M2+ and M4+ ions
Ans: (b)
Q.36. Is boric acid a protic acid? Explain.
Ans: No, boric acid is aprotic acid. It is Lewis acid and accepts OH-.
B(OH)3 + H2O → [B(OH)4]– + H+
Q.37. Explain what happens when boric acid is heated.
Ans: H3BO3
 HBO2
+ H2O
HBO2
+ H2O
2HBO2  B2O3 + H2O
B2O3 + H2O
Q.38. Describe the shapes of BF3 and BH4–. Assign the hybridisation of boron in these species.
Ans: BF3 is trigonal planar due to sp2 hybridisation.
BH4– is tetrahedral due to sp3 hybridisation.
Q.39. Write reactions to justify amphoteric nature of aluminium.
Ans: 2Al + 6HCl → 2AlCl3 + 3H2
2Al + 2NaOH + 2H2O → 2NaAlO2 + 3H2
Q.40. What are electron deficient compounds? Are BCl3 and SiCl4 electron deficient species? Explain.
Ans: Electron deficient compounds are those compounds in which octet of central atom is not complete. BCl3 is electron deficient because its octet is not complete. SiCl4 is not electron deficient because its octet is complete.
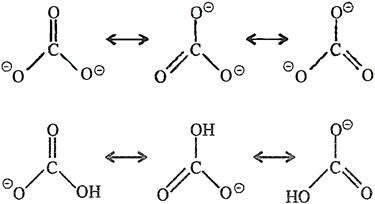
Q.41. Write the resonance structures of CO32– and HCO3–.
Ans:
Q.42. What is the state of hybridisation of carbon in
(i) CO32– (ii) diamond (iii) graphite?
Ans: (i) sp2 (ii) sp3 (iii) sp2
Q.43. Explain the difference in properties of diamond and graphite on the basis of their structures.
Ans: (i) Diamond is hard whereas graphite is soft.
(ii) Diamond is insulator whereas graphite is good conductor of heat and electricity.
Q.44. Rationalise the given statements and give chemical reactions:
(i) Lead (II) chloride reacts with Cl2 to give PbCl4.
(ii) Lead (IV) chloride is highly unstable towards heat.
(iii) Lead is known not to form an iodide, PbI4.
Ans: (i) PbCl2. + Cl2 → No reaction.
It is because Pb can also show +2 oxidation state more easily than +4 due to inert pair effect.
(ii) PbCl4 → PbCl2 + Cl2
It is because Pb2+ is more stable than Pb4+ due to inert pair effect.
(iii) It is because both Pb and I are very big in size, leading to very weak bond, therefore PbI4 does not exist. Secondly, Pb4+ is good oxidising agent and I– is good reducing agent. Hence, Pb4+ is reduced to Pb2+ and I– is oxidised to I2. Therefore, PbI4 does not exist.
Q.45. Suggest reasons why the B–F bond lengths in BF3 (130 pm) and BF4– (143 pm) differ.
Ans: In BF3, 'B' is sp2 hybridised and its structure is trigonal planar whereas in BF4–, 'B' is sp3 hybridised and there is one coordinate covalent bond, therefore, bond lengths are different.
Q.46. Write the members of group 14 that:
(i) forms the most acidic dioxide
(ii) is commonly found in +2 oxidation state
(iii) used as semiconductor
(iv) exhibit highest catenation tendency.
Ans: (i) Carbon (ii) Lead (iii) Silicon/Germanium (iv) Carbon
Q.47. What are allotropes? List two characteristic differences between diamond and graphite which are allotropes of carbon.
Ans: Allotropes are different forms of same element having different physical but similar chemical properties.
Two characteristic differences between diamond and graphite are:
(i) Diamond is hard while graphite is soft.
(ii) Diamond is a bad conductor of electricity while graphite is a good conductor of electricity.
Q.48. What happens when (Give reactions only):
(i) Silicon dioxide is treated with hydrogen fluoride.
(ii) Aluminium is treated with dil. NaOH.
Ans: (i) SiO2 + 4HF → SiF4 + 2H2O
In excess of HF, H2SiF6 is formed.
SiF4 + 2HF → H2SiF6 (Florosilicic acid)
(ii) 2Al + 2H2O + 2NaOH → 2NaAlO2 + 3H2
and it can form Na[Al(OH)4]
Q.49. (i) How will you explain higher stability of BCl3 as compared to TlCl3?
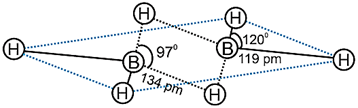
(ii) Draw the structure of diborane.
Ans: (i) In Tl3+ there is poor shielding f-orbitals which causes reluctance of the s-electrons to participate in bond formation leading to inert pair effect due to which Tl+ is more stable than Tl3+. In BCl3, there is back bonding i.e., lone pairs of 'Cl' are donated to 'B' to make it stable.
(ii)
Q.50.
 State with equations what
happens when borax is heated on a platinum wire loop and the resulting
transparent mass is heated with CoO in Bunsen burner.
State with equations what
happens when borax is heated on a platinum wire loop and the resulting
transparent mass is heated with CoO in Bunsen burner.
Ans: Na2B4O7.10H2O
 Na2B4O7
+ 10H2O
Na2B4O7
+ 10H2O
Na2B4O7
 2NaBO2 + B2O3
2NaBO2 + B2O3
Sodium metaborate Boric anhydride
transparent solid
This transparent solid is called borax bead. Whenever a coloured salt of Co2+, Cu2+, Ni2+ etc. is heated with borax bead, the salt decomposes to form the corresponding metal oxide which combines with B2O3 present in the bead to form coloured metaborates e.g.,
CoO + B2O3  Co(BO2)2
Co(BO2)2
Cobalt oxide Cobalt metaborate (blue)
Q.51. How are silicones prepared? Write its two uses.
Ans: Preparation of Silicones Methyl chloride (or any other alkyl or aryl chloride) reacts with silicon in presence of copper catalyst at 573 K, methyl substituted chlorosilanes are produced.
Hydrolysis of dialkyl chlorosilanes, followed by condensation polymerisation yields silicones.
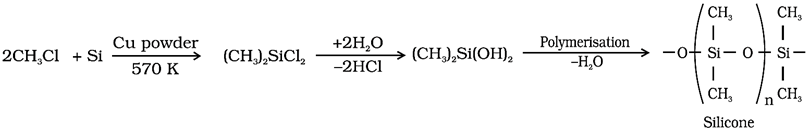
Uses of Silicones: Surgical implants; water proofing material.
Q.52. Discuss the pattern of variation in the oxidation states of
(i) B to Tl and (ii) C to Pb
Ans: (i) Tendency to show +1 oxidation state increases down the group due to inert pair effect.
(ii) Tendency to show +2 oxidation state increases down the group due to inert pair effect.
Q.53. How can you explain higher stability of BCl3 as compared to TlCl3?
Ans: It is because Tl+ is more stable than Tl3+ due to inert pair effect that is why TlCl3 is less stable than BCl3.
Q.54. Why does boron trifluoride behave as a Lewis acid?
Ans: It is because octet of boron is not complete. It is electron deficient therefore acts as Lewis acid.
Q.55. Consider the compounds, BCl3 and CCl4. How will they behave with water? Justify.
Ans: BCl3 + 3H2O → H3BO3 + 3HCl because BCl3 is covalent and electron. deficient. CCl4 does not react with water because C does not have vacant d-orbital and octet of carbon is complete.
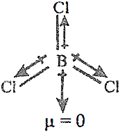
Q.56. If B–Cl bond has a dipole moment, explain why BCl3 molecule has zero dipole moment.
Ans: Although B–Cl bonds are polar bonds they get cancelled due to trigonal planar shape; net dipole moment is zero.
Q.57. Aluminium trifluoride is insoluble in anhydrous HF but dissolves on addition of NaF. Aluminium trifluoride precipitates out of the resulting solution when gaseous BF3 is bubbled through. Give reasons.
Ans: It is because HF is covalent compound and weak acid due to high bond dissociation energy whereas NaF is ionic compound.
3NaF + AlF3 → Na3[AlF6]
Na3[AlF6] + 3BF3(g) → AlF3 + 3Na+[BF4]–
Sodium tetrafluoroborate(III)
(Soluble complex)
Q.58. Suggest a reason as to why CO is poisonous.
Ans: It is because CO reacts with haemoglobin to form carboxyhaemoglobin which does not act as oxygen carrier.
Q.59. How is excessive content of CO2 responsible for global warming?
Ans: It is because CO2 absorbs infra-red radiations which cause heating effect.
Q.60. Explain structures of boric acid.
Ans:
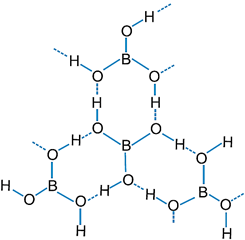
Q.61. What happens when
(i) borax is heated strongly?
(ii) boric acid is added to water?
(iii) aluminium is treated with dilute NaOH?
 (iv) BF3 is reacted with
ammonia?
(iv) BF3 is reacted with
ammonia?
Ans: (i) Na2B4O7.10H2O  Na2B4O7
+ 10H2O
Na2B4O7
+ 10H2O
Na2B4O7
 2NaBO2 + B2O3
2NaBO2 + B2O3
Sodium metaborate Boric anhydride
transparent solid
(ii) B(OH)3 + H2O → [B(OH)4]– + H+
(iii) 2Al + 2NaOH + 2H2O → 2NaAlO2 + 3H2
(iv) BF3 + NH3 → (H3N → BF3)
Q.62. Explain the following reactions:
(i) Silicon is heated with methyl chloride at high temperature in the presence of copper
(ii) Silicon dioxide is treated with hydrogen fluoride
(iii) CO is heated with ZnO
(iv) Hydrated alumina is treated with aqueous NaOH solution.
Ans: (i) Si + 2CH3Cl  Si(CH3)2Cl2
Si(CH3)2Cl2
(ii) SiO2 + 6HF → H2SiF6 + 2H2O
(iii) ZnO + CO → Zn + CO2
(iv) Al2O3.2H2O + 2NaOH → 2NaAlO2 + 3H2O
Q.63. Give reasons:
(i) Conc. HNO3 can be transported in aluminium container.
(ii) A mixture of dilute NaOH and aluminium pieces is used to open drain.
(iii) Graphite is used as lubricant.
(iv) Diamond is used as an abrasive.
(v) Aluminium alloys are used to make aircraft body.
(vi) Aluminium utensils should not be kept in water overnight.
(vii) Aluminium wire is used to make transmission cables.
Ans:
(i) It is because aluminium container coated with Al2O3 which makes it passive and does not react with conc. HNO3.
(ii) It is because it liberates hydrogen gas which exerts pressure to open drain.
(iii) Graphite is soft and slippery and has high melting point, therefore, acts as good lubricant.
(iv) It is hard substance, therefore, used as abrasive.
(v) It is light, tough and strong.
(vi) It forms layer of Al(OH)3 on the surface.
(vii) It is good conductor of electricity.
Q.64. Explain why is there a phenomenal decrease in ionization enthalpy from carbon to silicon.
Ans: It is due to increase in atomic size, therefore, ionisation energy decreases.
Q.65. How would you explain the lower atomic radius of Ga as compared to Al?
Ans: It is due to poor shielding effect of d-electrons in Ga.
Q.66. What are allotropes? Sketch the structure of two allotropes of carbon namely diamond and graphite. What is the impact of structure on physical properties of two allotropes?
Ans: Allotropy is property due to which an element exists in more than one form which differ in physical properties but have similar chemical properties. These different forms are called allotropes.
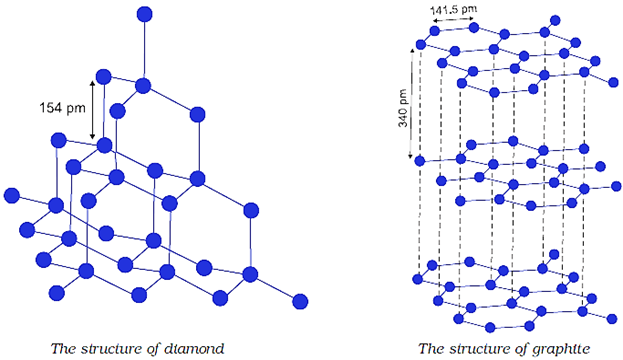
The structure of diamond makes it hard having high melting point and poor conductor of electricity. Structure of graphite makes it soft, slippery and good conductor of electricity.
Q.67. (i) Classify following oxides as neutral, acidic, basic or amphoteric:
CO, B2O3, SiO2, CO2, Al2O3, PbO2, Tl2O3
(ii) Write suitable chemical equations to show their nature.
Ans:
(i) Neutral: CO
Acidic: B2O3, CO2, SiO2
Basic: Tl2O3
Amphoteric Al2O3, PbO2
(ii) CO is neither acidic nor basic, it does not react with acid as well as base at room temperature.
B2O3 + 6NaOH → 2Na3BO3 + 3H2O
SiO2 + 2NaOH → Na2SiO3 + H2O
CO2 + 2NaOH → Na2CO3 + H2O
Al2O3 + 2NaOH → 2NaAlO2 + H2O
Al2O3 + 6HCl → 2AlCl3 + 3H2O
PbO2 + 4HCl → PbCl2 + Cl2 + 2H2O
PbO2 + 2NaOH → Na2PbO3 + H2O
Tl2O3 + 6HCl → 2TlCl3 + 3H2O
Q.68. In some of the reactions, thallium resembles aluminium, whereas in others it resembles with group I metals. Support this statement by giving some evidences.
Ans: In addition to +3, Tl also shows +1 oxidation state due to inert pair effect. Thus, it resembles with group I metals. Tl forms basic oxides like group I elements.
Q.69. Explain why BCl3 is monomer but BH3 exists as B2H6.
Ans: In BCl3, there is back bonding, i.e., lone pair of electrons is donated from chlorine to boron thus reducing its electron deficiency. In BH3, back bonding is not possible but H-bridges are possible, therefore, it exists as dimer.
Q.70. Why is BF3 planar molecule but NH3 is pyramidal?
Ans: In BF3, 'B' is sp2 hybridised whereas in NH3, 'N' is sp3 hybridised with one lone pair of electrons, therefore, it is pyramidal while BF3 is planar.
Q.71. Account for the following:
Why BF3 is less acidic than BCl3 though fluorine is more electronegative than chlorine?
Ans: BF3 is less acidic because back bonding is most effective due to 2p-2p overlapping whereas it is less effective in BCl3 due to 2p-3p overlapping.
Q.72. Arrange the following compounds in decreasing order of stability of +3 oxidation state. Give reason for your answer:
BCl3, AlCl3, GaCl3, InCl3, TlCl3
Ans: BCl3 > AlCl3 > GaCl3 > InCl3 > TlCl3 because of inert pair effect stability of +3 oxidation state decreases down the group.
Q.73. C and Si are always tetravalent but Ge, Sn, Pb show divalency.
Ans: Ge, Sn, Pb show divalency due to inert pair effect, e.g., Pb2+ is more stable than Pb4+.
Q.74. Which of the following is acidic and why?
SiO2, Al2O3, PbO2, SnO2
Ans: SiO2 is acidic oxide because Si is non-metal.
Q.75. Gallium has higher I.E. than Al. Explain.
Ans: It is due to poor shielding effect of d-electrons in Ga, effective nuclear charge increases as compared to Al, therefore, I.E. of Ga is higher than Al. Secondly, Ga is smaller than Al.
Q.76. Boron does not form [BF6]3– whereas [AlF6]3– exists, why?
Ans: It is because 'B' does not have vacant d-orbitals whereas 'Al' has vacant d-orbitals.
Q.77. If the starting material for the manufacture of silicones is RSiCl3, write the structure of the product formed.
Ans:
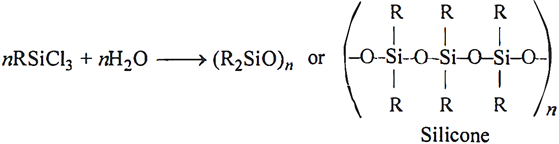
Q.78. Identify the compounds A, X and Z in the following reactions:
(i) A + 2HCl + 5H2O → 2NaCl + X
(ii) X  HBO2
HBO2
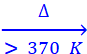 Z
Z
Ans:
(i) Na2B4O7 + 2HCl + 5H2O → 2NaCl + 4H3BO3
‘A’ ‘X’
(ii) H3BO3 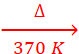 HBO2
HBO2  B2O3 + H2O
B2O3 + H2O
‘X’ ‘Z’
Q.79. Complete the following chemical equations:
(i) Z + 3LiAlH4 → X + 3LiF + 3AlF3
(ii) X + 6H2O → Y + 6H2
(iii) 3X + 3O2  B2O3 + 3H2O
B2O3 + 3H2O
Ans:
(i) 4BF3 + 3LiAlH4 → 2B2H6 + 3LiF + 3AlF3
‘Z’ ‘X’
(ii) B2H6 + 6H2O → 2H3BO3 + 6H2
‘X’ ‘Y’
(iii) 3B2H6 + 3O2
 B2O3 + 3H2O
B2O3 + 3H2O
‘X’
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
