Equilibrium
Practice Question Answer Set 1
Q.1. Give two important characteristics of chemical equilibrium.
AnswerHide Answer
Characteristics of chemical equilibrium are as follows
(i) Chemical equilibrium is dynamic in nature.
(ii) A catalyst does not alter the state of equilibrium.
Q.2. What is the equilibrium concentration of each of the substances in the equilibrium when the initial concentration of ICl was 0.78 M?
2ICl(g) → I2(g) + Cl2(g); Kc = 0.14
AnswerHide Answer
2ICl(g) ⇌ I2(g) + Cl2(g); Kc = 0.14
Initial conc. 0.78 0 0
Equi. conc. 0.78 –2x x x
Applying law of chemical equilibrium,
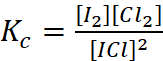
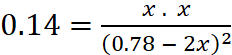
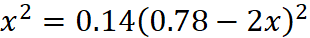
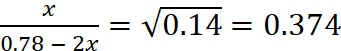
or x = 0.292 – 0.748x
or 1.748x = 0.292 or x = 0.167
Hence at equilibrium, [I2] = [Cl2] = 0.167 M
[ICl] = 0.78 – 2 × 0.167 = 0.446 M
Q.3. Give relation
between [A] and [B] for the stage of half completion of the reaction 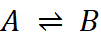 .
.
At the stage of half completion of reaction 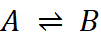 ,
,
[A] = [B].
Q.4. The pH of a sample of vinegar is 3.76. Calculate the concentration of hydrogen ions in it.
AnswerHide Answer
We know that pH = – log[H+]
∴ [H+] = antilog[–pH] = antilog(–3.76)
= 1.738 × 10–4 M
Q.5. A mixture of 1.57 mol of N2, 1.92 mol of H2 and 8.13 mol of NH3 is introduced into a 20 L reaction vessel at 500 K. At this temperature, the equilibrium constant, Kc for the reaction
N2(g) + 3H2(g) ⇌ 2NH3(g)
is 1.7 × 102.
Is the reaction mixture at equilibrium? If not, what is the direction of the net reaction?
AnswerHide Answer
The reaction is
N2(g) + 3H2(g) ⇌ 2NH3(g)
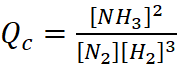
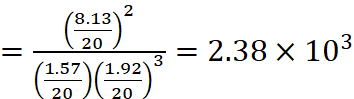
As Qc ≠ Kc, the reaction mixture is not in equilibrium.
As Qc > Kc, the net reaction will be in the backward direction.
Q.6. Explain why pure liquids and solids can be ignored while writing the equilibrium constant expression.
AnswerHide Answer
For the concentration of pure solid or pure liquid,
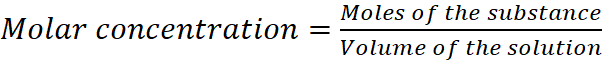
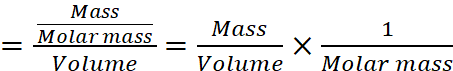
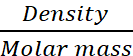
Since density of pure solid or liquid is constant at constant temperature and molar mass is also constant therefore, their molar concentrations are constant and are included in the equilibrium constant.
Q.7. Conjugate acid of a weak base is always stronger. What will be the decreasing order of basic strength of the following conjugate bases?
OH–, RO–, CH3COO–, Cl–
AnswerHide Answer
Conjugate acids of given bases are H2O, ROH, CH3COOH, HCl.
Their acidic strength is in the order
HCl > CH3COOH > H2O > ROH
Hence, basic strength is in the order
RO– > OH– > CH3COO– > Cl–
Q.8. Classify the following species into Lewis acids and Lewis bases and show how these act as Lewis acid/base:
(a) OH– (b) F– (c) H+ (d) BCl3.
AnswerHide Answer
(a) OH– : OH– is a Lewis base because it can donate lone pair of electrons.
(b) F– : F– is a Lewis base because it can donate lone pair of electrons.
(c) H+ : H+ is a Lewis acid because it can accept lone pair of electrons.
(d) BCl3 : BCl3 is a Lewis acid because it is electron deficient and can accept a lone pair of electrons.
Q.9. Calculate the pH of a solution formed by mixing equal volumes of two solutions A and B of a strong acid having pH = 6 and pH = 4 respectively.
AnswerHide Answer
pH of solution A = 6
[H+] = 10–6 mol L–1
pH of solution B = 4
[H+] = 10–4 mol L–1
On mixing one litre of each solution
Total volume = 1 L + 1 L = 2 L
Total amount of H+ in 2 L solution formed by mixing solutions A and B
= 10–6 + 10–4 mol
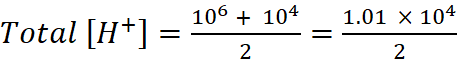
= 5 × 10–5 mol L–1
pH = –log[H+] = –log(5 × 10–5)
= –log5 – (–5 log10) = –log 5 + 5
= 5 – log 5 = 5 – 0.6990
= 4.3010 = 4.3
Q.10. What will be the conjugate bases for the Bronsted acids: HF, H2SO4 and HCO3–?
AnswerHide Answer
|
Bronsted acid |
Conjugate base |
|
HF |
F– |
|
H2SO4 |
HSO4– |
|
HCO3– |
CO32– |
