Important Inorganic Reactions – Set 1
1. Pb(NO3)2 + 2NaOH → Pb(OH)2 + 2NaNO3
2. Zn(OH)2 + 2NaOH → Na2ZnO2 + 2H2O
3. 2Na[Al(OH)4] + CO2 → 2Al(OH)3 + Na2CO3
4. CuSO4 + 2NaOH(excess) → Cu(OH)2 + Na2SO4
5. Fe(OH)3 + NaOH(excess) → No reaction
6. Mg(OH)2 + 2HCl → MgCl2 + 2H2O
7. Mn(NO3)2 + 2NaOH → Mn(OH)2 + 2NaNO3
8. CH3COOAg + HNO3 → AgNO3 + CH3COOH
9. Hg(NO3)2 + NH3(soln. ) → HgO.sHgNH2NO3
10. Cu(OH)2 + 4NH3(soln. ) → [Cu(NH3)4]2 + + 2OH−
11. CaC2O4 + CH3COOH → No reaction
12. BaC2O4 + 2CH3COOH → Ba(CH3COO)2 + H2C2O4
13. Fe(CN)2 + 4KCN → K4[Fe(CN)6]
14. SrC2O4 + 2HCl → SrCl2 + H2C2O4
15. Fe(CN)3 + KCN → K3Fe(CN)6
16. CaSO3 + SO2 + H2O → Ca(HSO3)2
17. K4[Fe(CN)6] + ZnSO4 → Zn2[Fe(CN)6]
18. 3PbS + 8HNO3(dil. ) → 3Pb(NO3)2 + 3S + 2NO
19. K4[Fe(CN)6] + 2CuSO4 → Cu2[Fe(CN)6]
2O. MnS + 2HCl → MnCl2 + H2S
21. AgCl + 2KCN → K[Ag(CN)2] + KCl
22. HgS + Na2S → Na2[HgS2]
23. CuSO4 + 2KCN → CuCN + (CN)2 + K2SO4
24. FeS + 2HCl → FeCl2 + H2S
25. Cd(CN)2 + 2KCN → K2[Cd(CN)4]
26. 2AgF + MgCl2 → MgF2 + 2AgCl
27. Pb(NO3)2 + 2KI → PbI2 + 2KNO3
28. PbCl2 + Hot water → Pb2 + (aq. ) + 2Cl−(aq. )
29. HgI2 + KI → K2[HgI4]
30. AgI + 2Na2S2O3 → Na3[Ag(S2O3)2] + NaI
31. CuSO4 + 2KI → CuI + 1/2I2 + K2SO4
32. KNO2 + AgF → AgNO2 + KF
33. BaSO4 + Na2CO3 → No reaction
34. FeCl3 + Na3PO4 → FePO4 + 3NaCl
35. BaSO4 + dil. HCl(Excess) → No reaction
36. 2AgNO3 + Na2C2O4 → Ag2C2O4 + 2NaNO3
37. 2BaCrO4 + 4HCl → 2BaCl2 + H2Cr2O7 + H2O
38. PbCrO4 + 4NaOH(Excess) → Na2[Pb(OH)4]
39. BaCrO4 + CH3COOH(Excess) → No reaction
40. PbCl2 + H2SO4
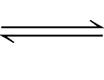 PbSO4 + 2HCl
PbSO4 + 2HCl
41. Ba(NO3)2 + Na2SO4 → BaSO4 + 2NaNO3
42. Pb(NO3)2 + H2SO4 → PbSO4 + 2HNO3
43. SrCrO4 + 2CH3COOH(Excess) → Sr(CH3COO)2 + H2Cr2O7
44. MCrO4(M2+ = Ba2+, Pb2+) + CH3COOH → No dissolution
45. CaCl2 + Na2C2O4 → CaC2O4 + 2NaCl
46. CaSO4 + Pb(NO3)2 → PbSO4 + Ca(NO3)2
47. Hg2(NO3)2 + NH3(solution) → Hg + HgO·HgNH2NO3
48. BaCO3 + 2HCl → BaCl2 + CO2 + H2O
49. AlCl3 + 3NaOH → Al(OH)3 + 3NaCl
50. BaCO3 + CO2 + H2O → Ba(HCO3)2
51. ZnS + 2HCl → ZnCl2 + H2S
52. NiCl2 + 2dmg 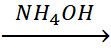 Ni(dmg)2
Ni(dmg)2
53. CaCl2 + Na2SO4 → No reaction
54. BaCO3 + 2CH3COOH → Ba(CH3COO)2 + CO2 + H2O
55. Na2S2O3 + BaCl2 → BaS2O3 + 2NaCl
56. Ba(CH3COO)2 + K2CrO4 → BaCrO4 + 2CH3COOK
57. 3AgNO3 + Na3PO4 → Ag3PO4 + 3NaNO3
58. Ag2CO3 + 2HCl → AgCl + CO2 + H2O
59. BaSO3 + H2SO4 → BaSO4 + SO2 + H2O
60. HgS + conc. HNO3 → No dissolution
61. Sr(CH3COO)2 + Ag2SO4 → 2CH3COOAg + SrSO4
62. Ca(OH)2 + 2HF → CaF2 + 2H2O
63. Ca(OH)2 + CO2 → CaCO3 + H2O
64. CaSO3 + H2SO4 → CaSO4 + SO2 + H2O
65. Ca(OH)2 + SO2 → CaSO3 + H2O
66. Na2SO3 + BaCl2 → BaSO3 + 2NaCl
67. Pb(CH3COO)2 + H2S → PbS + 2CH3COOH
68. NaCl + AgNO3 → AgCl + NaNO3
69. HgI2 + 2KI → K2[HgI4]
70. PbO2 + HNO3(dil.) → No dissolution
71. PbO2 + HNO3(Conc.) → Pb(NO3)2 + H2O + [O]
72. K2[Cd(CN)4] + H2S → CdS + 2KCN + 2HCN
73. Pb(CH3COO)2 + Na2CrO4 → PbCrO4 + 2CH3COONa
74. NaBr + AgNO3 → AgBr + NaNO3
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
