Class 12 Chemistry
Chapter 3 – Electrochemistry
Magical Series Assignment 3
Q.1. For the cell reaction,
Ni(s) | Ni2+(aq) || Ag+(aq) | Ag(s)
Calculate the equilibrium constant at 25°C. How much maximum work would be obtained by operation of this cell?
EoNi2+/Ni = –0.25 V and EoAg+/Ag = 0.80 V
Q.2. Calculate the standard cell potential of the galvanic cell in which the following reaction takes place:
Fe2+(aq) + Ag+ (aq) → Fe3+(aq) + Ag(s)
Calculate the ∆rGo and equilibrium constant of the reaction.
(EoAg+/Ag = 0.80 V; EoFe3+/Fe2+ = 0.77 V)
Q.3. Calculate the emf of following cell at 298 K:
Mg(s) | Mg2+(0.1 M) || Cu2+(0.01 M) | Cu(s)
[Given: Eocell = +2.71 V, 1 F = 96500 C mol–1]
Q.4. Estimate the minimum potential difference needed to reduce Al2O3 at 500 °C. The Gibbs energy change for the decomposition reaction,
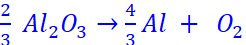 is 960 kJ (F = 96500 C mol–1)
is 960 kJ (F = 96500 C mol–1)
Q.5. Calculate the emf of the following cell at 298 K:
Fe(s) | Fe2+(0.001 M) || H+(l M) | H2(g)(1 bar), Pt(s)
(Given Eocell = +0.44 V)
Q.6. Calculate the emf of the following cell at 25°C:
Ag(s) | Ag+(10–3 M) || Cu2+(10–1 M) | Cu(s)
Given: Eocell = +0.46 V and log 10" = n.
Q.7. In the button cell, widely used in watches, the following reaction takes place.
Zn(s) + Ag2O(s) + H2O(l) → Zn2+(aq) + 2Ag(s) + 2OH–(aq)
Determine Eo and ∆Go for the reaction.
[Given: EoAg+/Ag = +0.80 V, EoZn2+/Zn = –0.76 V]
Q.8. A voltaic cell is set up at 25°C with the following half cells:
Al/Al3+(0.001 M) and Ni/Ni2+(0.50 M)
Write an equation for the reaction that occurs when the cell generates an electric current and determine the cell potential.
EoNi2+/Ni = –0.25 V and EoAl3+/Al = –1.66 V. (log 8 × 10–6 = –5.09)
Q.9. The cell in which the following reaction occurs:
2Fe3+(aq) + 2I–(aq) → 2Fe2+(aq) + I2(s)
has Eocell = 0.236 V at 298 K. Calculate the standard Gibbs energy and the equilibrium constant of the cell reaction.
(Antilog of 6.5 = 3.162 × 106; of 8.0 = 10 × 108; of 8.5 = 3.162 × 108)
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
