Chapter 1 – Basic Concepts of Chemistry
Important Questions Answers Set 2
Q.1.
Boron
occurs in nature in the form of two isotopes, 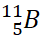 and
and 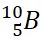 , in ratio of 81% and 19%
respectively. Calculate its average atomic mass.
, in ratio of 81% and 19%
respectively. Calculate its average atomic mass.
Q.2. If 2 litres of N2 is mixed with 2 litres of H2 at a constant temperature and pressure, then what will be the volume of NH3 formed?
Q.3. How many atoms are present in 1 ml of NH3 at STP?
Q.4. Which of these weighs most?
(i) 32 g of oxygen
(ii) 2 g atom of hydrogen
(iii) 0.5 mole of Fe
(iv) 3.01 × 1023 atoms of carbon
Q.5. Calculate the number of moles of NaOH in 27 cm3 of 0.15 M NaOH solution.
Q.6. Calculate the number of nm in 5839 A.
Q.7. How many gram atoms are there in 8.0 g of S?
Q.8. 0.5 mole each of H2S and SO2 mixed together in a reaction flask, react according to equation:
2H2S + SO2 → 2H2O + 3S
Calculate the number of moles of 'S' formed.
Q.9. If 6.023 × 1023 molecules of N2 react completely with H2 according to the equation:
N2(g) + 3H2(g) → 2NH3(g),
then calculate the number of molecules of NH3 formed. [Ans. 1.204 × 1024]
Q.10. Calculate the volume of 34 g of NH3 at STP.
Q.11.
Calculate
the mass of ferric oxide that will be obtained by complete oxidation of 2 g of
Fe.
[Atomic weights of Fe = 56 u, O = 16 u]
Q.12. How are 0.50 mol Na2CO3 and 0.50 M Na2CO3 different?
Q.13. How many atoms of calcium are there in 2 g of Ca? (At. mass of Ca = 40 u)
Q.14. Calculate the mass of 112 cm3 of hydrogen gas at STP. (Atomic mass of H = 1 u) [Ans. 0.01 g]
Q.15. Calculate the number of atoms present in 1.4 g of N2 molecule.
Q.16. What will be the mass of one 12C atom in g?
Q.17. Which one of the following will have the largest number of atoms?
(i) 1 g Au(s)
(iii) 1 g Li(s)
(ii) 1 g Na(s)
(iv) 1 g of Cl2(g)
Q.18. How many significant figures should be present in the answer of the following calculations?
(i)
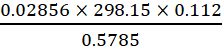
(ii) 5 × 5.364
(iii) 0.0125 + 0.7864 + 0.0215
Answers
A.1:
Average atomic weight = 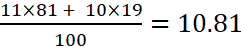
A.2: N2(g) + 3H2(g) → 2NH3(g)
1 L of N2 will react with 3 L of H2.
2 L of N2 will react with 6 L of H2 but we have only 2 L of H2, therefore, H2 is limiting reactant.
3 L of H2 gives 2 L of NH3
Þ 2 L of H2 gives 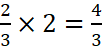 =
1.33 L of NH3
=
1.33 L of NH3
A.3: 22400 ml of NH3 contains = 4 × 6.022 × 1023 atoms [ as NH3 contains 4 atoms]
l ml of NH3 contains = 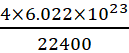 = 1.07 × 1020 atoms
= 1.07 × 1020 atoms
A.4: (i) 32 g of oxygen weighs most.
(ii) 2 g atom of H2 = 2 g
(iii) 0.5 mole of Fe = 0.5 × 56 = 28 g
(iv) 3.01 × 1023 atoms of C =  mol =
mol = 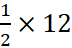 = 6 g
= 6 g
A.5:
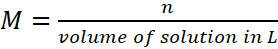
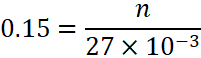
n = 0.00405 moles
A.6: l A = 10−10 m, l nm = 10−9 m
Þ 5839 A = 583.9 nm
A.7: 32 g of S = l g atom
Þ 8
g of S = 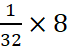 = 0.25 g atom
= 0.25 g atom
A.8: 2H2S + SO2 → 2H2O + 3S
2 moles of H2S combine with l mole of SO2 to give 3 moles of S.
l mole of H2S combines with 0.5 mole of SO2 to
give  moles of S.
moles of S.
Therefore, 0.5 mole of H2S combines with 0.25 mole of SO2
to give 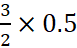 = 0. 75 moles of S.
= 0. 75 moles of S.
A.9: N2(g) + 3H2(g) → 2NH3(g)
6.023 × 1023 molecules of N2 react completely with H2 to give 2 × 6.023 × 1023 molecules of NH3
= 1.204 × 1024 molecules
A.10: Mol. wt. of NH3 = 14 + 3 = 17 g mol−1
l mole of NH3 = 17 g of NH3 has volume = 22.4 L
at STP, 34 g of NH3 = 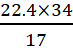 = 44.8 L
= 44.8 L
A.11: 4Fe + 3O2 → 2Fe2O3
4 × 56 g of Fe gives 2 × 160 g of Fe2O3.
2 g of Fe gives 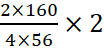 = 2.857 g of Fe2O3.
= 2.857 g of Fe2O3.
A.12: 0.50 mol of Na2CO3 = 0.50 × 106 = 53 g of Na2CO3
0.50 M Na2CO3 means 0.5 mol of Na2CO3, i.e.
53 g of Na2CO3 has been dissolved in 1 litre of solution.
A.13: 1 mole of Ca = 40 g = 6.022 × 1023 atoms
Þ 2
g of Ca = 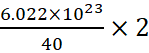 = 3.011 × 1022
= 3.011 × 1022
A.14: Since 22400 cm3 of hydrogen at STP weighs = 2 g
Therefore, 112 cm3 of hydrogen at STP weighs
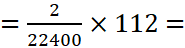 0.01 g
0.01 g
A.15: 28 g of N2 molecules contain 2 × 6.023 × 1023 atoms
Hence, 1.4 g of N2 molecules contain
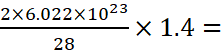 6.023 × 1022 atoms
6.023 × 1022 atoms
A.16: 1 mole of C = 12 g
1 mole of C contains = 6.023 × 1023 atoms
6.023 × 1023 atoms of carbon weigh = 12 g
l atom of carbon weighs =  = 1.99 × 10−23 g
= 1.99 × 10−23 g
A.17: 1 g of Li will have the largest number of atoms because it has lowest atomic mass.
A.18: (i) 3 (ii) 4 (iii) 4
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
