Q.1. A reaction is of second order with respect to its reactant. How will its reaction rate be affected if the concentration of the reactant is
(i) doubled
(ii) reduced to half ?
Q.2. What do you understand by the 'order of a reaction'? Identify the reaction order from each of the following units of reaction rate constant:
(i) L−1 mol s−1
(ii) L mol−1 s−1
Q.3. Distinguish between 'rate expression' and 'rate constant' of a reaction.
Q.4. What do you understand by the rate law and rate constant of a reaction? Identify the order of a reaction if the units of its rate constant are:
(i) L−1 mol s−1
(ii) L mol−1 s−1
Q.5. Express clearly what do you understand by 'rate expression' and 'rate constant' of a reaction.
Q.6. Explain the term 'order of reaction'. Derive the unit for first order rate constant.
Q.7. A reaction is second order in A and first order in B.
(i) Write the differential rate equation.
(ii) How is the rate affected on increasing the concentration of A three times?
(iii) How is the rate affected when the concentration of both A and B are doubled?
Q.8. The reaction, N2(g) + O2(g) → 2NO(g) contributes to air pollution whenever a fuel is burnt in air at a high temperature. At 1500 K, equilibrium constant K for it is 1.0 × 10−5. Suppose in a case [N2] = 0.80 mol L−1 and [O2] = 0.20 mol L−1 before any reaction occurs. Calculate the equilibrium concentrations of the reactants and the product after the mixture has been heated to 1500 K.
Q.9.
(i) A reaction is first order in A and second order in B.
(a) Write differential rate equation.
(b) How is rate affected when concentration of B is tripled?
(c) How is rate affected when concentration of both A and B is doubled?
(ii) What is molecularity of a reaction?
Q.10. The following results have been obtained during the kinetic studies of the reaction :
2A + B → C + D
|
Experiment |
[A] |
[B] |
Initial rate of formation of D |
|
1. |
0.1 M |
0.1 M |
6.0 × 10−3 M min−1 |
|
2. |
0.3 M |
0.2 M |
7.2 × 10−2 M min−1 |
|
3. |
0.3 M |
0.4 M |
2.88 × 10−1 M min−1 |
|
4. |
0.4 M |
0.1 M |
2.40 × 10−2 M min−1 |
Calculate the rate of formation of D when
[A] = 0.5 mol L−1 and
[B] = 0.2 mol L−1.
Q.11. The thermal decomposition of HCO2H is a first order reaction with a rate constant of 2.4 × 10−3 s−1 at a certain temperature. Calculate how long will it take for three−fourth of initial quantity of HCO2H to decompose. (log 0.25 = − 0.6021)
Q.12. In a first order reaction, the concentration of the reactant is reduced from 0.6 mol L−1 to 0.2 mol L−1 in 5 minutes. Calculate the rate constant of the reaction.
Q.13. Show that for a first order reaction, the time required for half the change (half−life period) is independent of initial concentration.
Q.14. Following data are obtained for the reaction:
N2O5 → 2NO2 + ½O2
|
t/s |
0 |
300 |
600 |
|
[N2O5]/ mol L−1 |
1.6 × 10−2 |
0.8 × 10−2 |
0.4 × 10−2 |
(a) Show that it follows first order reaction.
(b) Calculate the half−life.
(Given: log 2 = 0.3010, log 4 = 0.6021)
Q.15. A first order reaction takes 20 minutes for 25% decomposition. Calculate the time when 75% of the reaction will be completed.
(Given : log 2 = 0.3010, log 3 = 0.4771, log 4 = 0.6021)
Q.16. Define the half−life period of a reaction.
Q.17. If half−life period of a first order reaction is x and 3/4th life period of the same reaction is y, how are x and y related to each other?
Q.18. For a chemical reaction R → P, the variation in the concentration [R] vs. time (t) plot is given as
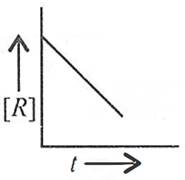
(i) Predict the order of the reaction.
(ii) What is the slope of the curve?
Q.19. Define half−life of a reaction. Write the expression of half−life for
(i) zero order reaction
(ii) first order reaction.
Q.20. For a first order reaction, show that time required for 99% completion is twice the time required for the completion of 90% of reaction.
Online Tuitions and Self-Study Courses for Grade 6 to 12 & JEE / NEET
