LOTUS VALLEY INTERNATIONAL SCHOOL, NOIDA
ANNUAL EXAMINATION (2020 -2021)
SUBJECT- CHEMISTRY
Duration: 3 hours Max. Marks: 70
General Instructions
a) There are 33 questions in this question paper. All questions are compulsory.
b) Section A: Q. No. 1 to 2 are case-based questions having four MCQs or Reason Assertion type based on given passage each carrying 1 mark.
c) Section A: Question 3 to 16 are MCQs and Reason Assertion type questions carrying 1 mark each.
d) Section B: Q. No. 17 to 25 are short answer questions and carry 2 marks each.
e) Section C: Q. No. 26 to 30 are short answer questions and carry 3 marks each.
f) Section D: Q. No. 31 to 33 are long answer questions carrying 5 marks each.
g) There is no overall choice. However, internal choices have been provided.
h) Use of calculators and log tables is not permitted.
SECTION - A (OBJECTIVE TYPE)
1. Read the passage given below and answer the following questions:
The mole concept is a convenient method of expressing the amount of a substance. Any measurement can be broken down into two parts – the numerical magnitude and the units that the magnitude is expressed in. For example, 60 grams of carbon is expressed as 5 moles. The mole (symbol: mol) is the unit of measurement for amount of substance in the International System of Units (SI). A mole of a substance or a mole of particles is defined as the amount containing exactly 6.02214076 × 1023 particles, which may be atoms, molecules, ions, or electrons. In short, 1 mol contains 6.02214076 × 1023 of the specified particles. Number of molecules is also related to volume of gases under specific conditions of Temperature and Pressure.
i. Which of the following has maximum number of moles?
(a) 4 grams of dihydrogen
(b) 4 grams of dinitrogen
(c) 4 grams of ozone
(d) 4 grams of dioxygen
ii. Which of the following occupies maximum volume at STP?
(a) 11 grams of CO2
(b) 2 grams of H2
(c) 16 grams of O2
(d) 14 grams of N2
iii. Which of the following is incorrect statement?
(a) 1 mole carbon dioxide contains 12 grams of carbon and 32 grams of oxygen.
(b) 1 mole carbon dioxide contains 1 grams of carbon and 2 grams of oxygen.
(c) 1 mole carbon dioxide contains 1 grams of carbon and 1 grams of oxygen.
(d) 1 mole carbon dioxide contains 12 grams of carbon and 8 grams of oxygen.
iv. Consider the following reaction: H2 + Cl2 → 2HCl
The correct statement with reference to above equation is?
(a) 2 grams of hydrogen reacts with 2 grams of Chlorine.
(b) 1 grams of hydrogen reacts with 2 grams of Chlorine.
(c) 2 grams of hydrogen reacts with 1 grams of Chlorine.
(d) 1 mole of hydrogen reacts with 1 mole of Chlorine.
OR
1 mole of any gas at STP occupies:
(a) 22.4 litre
(b) 44.8 litre
(c) 11.2 litre
(d) 1 litre
2. Read the passage given below and answer the following questions:
In Thermodynamics, whole universe is divided into two parts: System and Surrounding. System is any specified portion of universe under investigation.
System + Surroundings = Universe
There are three types of system: Open system, Closed System and Isolated system. Thermodynamic Process is an operation by which a system undergoes a change from one state to another. It may be carried out reversibly and irreversibly depending upon the speed at which it is carried out. Thermodynamic properties if depends only on initial and final states of the system are termed as State functions, they are also classified as Extensive and Intensive properties based on different parameters.
In the following questions, a statement of assertion followed by a statement of reason is given.
Choose the correct answer out of the following choices.
a) Assertion and reason both are correct statements and reason is correct explanation for assertion.
b) Assertion and reason both are correct statements but reason is not correct explanation for assertion.
c) Assertion is correct statement but reason is wrong statement.
d) Assertion is wrong statement but reason is correct statement.
i. Assertion: Work done by the system is not a state function.
Reason: Work done by a system depends upon the final and initial state of the system.
ii. Assertion: For cyclic process, ∆S, ∆G and ∆H are zero.
Reason: Because ∆S, ∆G, ∆H are state functions.
iii. Assertion: The slope of an irreversible process is lesser than that of reversible process, when P and V is drawn in Y-axis and X-axis respectively.
Reason: Work done in reversible process is more than the work done in Irreversible process.
OR
Assertion: Combustion of all organic compounds is an exothermic reaction.
Reason: The enthalpies of all elements in their standard state are zero.
iv. Assertion: Heat absorbed during the isothermal expansion of an ideal gas against vacuum is zero.
Reason: Heat absorbed during all isothermal processes is zero.
Following questions (No. 3-11) are multiple choice questions carrying 1 mark each:
3. Atomic number of the elements of group 13 of the periodic table are:
(a) 3, 11, 19, 37
(b) 5, 13, 21, 39
(c) 7, 15, 31, 49
(d) 5,13,31,49
4. The hybrid state of B in BF4– is:
(a) sp2
(b) sp
(c) sp3
(d) sp3d
OR
Shape of a molecule of BrF5 is:
(a) Tetrahedral
(b) Octahedral
(c) Planar
(d) Square pyramidal
5. Dipole moment of NF3 is smaller than:
(a) NH3
(b) CO2
(c) BF3
(d) CCl4
6. Representative elements belong to:
(a) s and p block
(b) d block
(c) d and f block
(d) f block
OR
Noble gases are present in:
(a) s block
(b) p block
(c) d block
(d) f block
7. The bond order of NO molecule is:
(a) 1.5
(b) 2
(c) 2.5
(d) 3.0
OR
Bond order of He2 molecule is:
(a) 1.5
(b) 2
(c) 2.5
(d) 0
8. Equal weights of Ethane and Hydrogen are mixed in an empty container at 25 ˚C. The fraction of total pressure exerted by Hydrogen is:
(a) 1 : 1
(b) 1 : 2
(c) 1 : 16
(d) 15 : 16
9. The pH of neutral water at 25°C is 7.0. As the temperature increases, ionisation of water increases, however, the concentration of H+ ions and OH– ions are equal. What will be the pH of pure water at 60°C?
(a) Equal to 7.0
(b) Greater than 7.0
(c) Less than 7.0
(d) Equal to zero
OR
An aqueous solution of NaCl is:
(a) Neutral
(b) Acidic
(c) Alkaline
(d) Depends upon the temperature
10. In which of the following compounds, the carbon marked with asterisk is most electronegative?
(a) CH3 – CH2 – *CH2 –CH3
(b) CH3 – *CH = CH – CH3
(c) CH3 – CH2 – C ≡ *CH
(d) CH3 – CH2 – CH = *CH2
11. In going from left to right in a period:
(a) the basic nature of the oxides increases
(b) acidic nature of the oxides increases.
(c) the acidic nature of the oxides decreases
(d) no change in the nature of oxides.
In the following questions (Q. No. 12 - 16) a statement of assertion followed by a statement of reason is given.
Choose the correct answer out of the following choices.
a) Assertion and reason both are correct statements and reason is correct explanation for assertion.
b) Assertion and reason both are correct statements but reason is not correct explanation for assertion.
c) Assertion is correct statement but reason is wrong statement.
d) Assertion is wrong statement but reason is correct statement.
12. Assertion: The IUPAC name of following compound is cycloocta-1,3,5,7-tetraene.
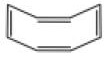
Reason: Above compound is also known as Naphthalene.
13. Assertion: Among isomeric pentanes, 2, 2-dimethylpropane has lowest boiling point.
Reason: Branching does not affect the boiling point.
14. Assertion: All the carbon atoms in H2C = C = CH2 are sp2 hybridised. Reason: In this molecule all the carbon atoms are attached to each other by double bonds.
OR
Assertion: Pent- 1- ene and Pent- 2- ene are position isomers.
Reason: Position isomers differ in the position of functional group or a substituent.
15. Assertion: Energy of resonance hybrid is equal to the average of energies of all canonical forms.
Reason: Resonance hybrid cannot be presented by a single structure.
16. Assertion: Boron has a smaller first ionisation enthalpy than beryllium. Reason: The penetration of a 2s electron to the nucleus is more than the 2p electron hence 2p electron is more shielded by the inner core of electrons than the 2s electrons.
SECTION B
The following questions, Q. No 17 – 25 are short answer type and carry 2 marks each.
17. An element ‘X’ has atomic number 13:
(a) Write its electron configuration.
(b) State the group to which ‘X’ belongs?
(c) Is ‘X’ a metal or a non-metal?
(d) Write the formula of its bromide.
OR
Identify the group of an element having atomic number 119. Also predict the outermost electronic configuration.
18. Calculate the total number of angular nodes and radial nodes present in 3p orbital.
OR
Out of electron and proton which one will have, a higher velocity to produce matter waves of the same wavelength? Explain it.
19. A vessel of 120 mL capacity contains a certain amount of gas at 35 °C and 1.2 bar pressure. The gas is transferred to another vessel of volume 180 mL at 35 °C. What would be its pressure?
20. Enthalpy of combustion of carbon to CO2 is –393.5 kJ mol–1. Calculate the heat released upon formation of 35.2 g of CO2 from carbon and dioxygen gas.
21. For the reaction at 298 K, 2A + B → C
ΔH = 400 kJ mol–1 and ΔS = 0.2 kJ K–1 mol–1. At what temperature will the reaction become spontaneous considering ΔH and ΔS to be constant over the temperature range.
22. Explain why pure liquids and solids can be ignored while writing the equilibrium constant expression?
23. Arrange the following in the increasing order of bond length mentioned.
i) CH3−CH3, CH2=CH2, HC≡CH (C−C bond)
ii) CH3−CH3, CH2=CH2, HC≡CH (C−H bond)
24. i) Sodium salt of which acid will be needed for the preparation of propane? Write chemical equation for the reaction.
ii) Write down the balanced chemical reaction of preparing Prop-1-ene from appropriate vicinal dibromide using Zn metal.
OR
Complete the following reactions:
i. CH3CH=CH2 +
H2O 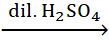
ii. HC≡CH + H2O 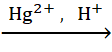
iii. CH3CH=CH2 + HBr →
iv. CH3−C≡C−CH3
+ H2 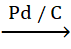
25. Write structures of different isomers corresponding to the 3rd member of alkyne series.
Also write the IUPAC names of all the isomers.
SECTION C
Q. No 26 -30 are Short Answer Type II carrying 3 mark each.
26. Chlorophyll present in green leaves of plants absorbs light at 4.620 × 1014 Hz. Calculate the wavelength of radiation in nanometre. Which part of the electromagnetic spectrum does it belong to?
OR
Calculate the energy and frequency of the radiation emitted when an electron jumps from n = 3 to n = 2 in a hydrogen atom.
27. A mixture of dihydrogen and dioxygen at one bar pressure contains 20% by weight of dihydrogen. Calculate the partial pressure of dihydrogen.
OR
At 25°C and 760 mm of Hg pressure a gas occupies 600 mL volume. What will be its pressure at a height where temperature is 10°C and volume of the gas is 640 mL.
28. Balance the reaction given below using Half reaction method:
S2O32– + 2Br2 + 5H2O → 2SO42– + 4Br– + H+
29. Explain Inductive effect. Arrange the following carbocations in the increasing order of stability:
i) 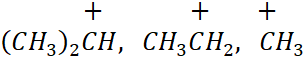
ii) 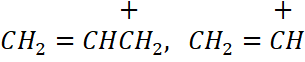
30. An alkene ‘A’ contains three C – C, eight C – H σ bonds and one C – C π bond. ‘A’ on ozonolysis gives two moles of an aldehyde of molar mass 44 u. Write IUPAC name of ‘A’, also write all the reactions involved.
SECTION D
Q. No 31 to 33 are long answer type carrying 5 marks each.
31. (i) Define Dipole Moment. What is its significance?
(ii) Represent diagrammatically the bond moments and the resultant dipole moment in CO2, NH3 and CHCl3.
OR
Explain briefly about Molecular Orbital Theory. Use the molecular orbital energy level diagram to show that N2 would be expected to have a triple bond, F2, a single bond and Ne2 no bond.
32. Which of the following reactions will get affected by increasing the pressure? Also, mention whether change will cause the reaction to go into forward or backward direction.
(i) COCl2(g) ⇋ CO(g) + Cl2(g)
(ii) CH4(g) + 2S2(g) ⇋ CS2(g) + 2H2S(g)
(iii) CO2(g) + C(s) ⇋ 2CO(g)
(iv) 2H2(g) + CO(g) ⇋ CH3OH(g)
(v) CaCO3(s) ⇋ CaO(s) + CO2(g)
OR
(i) The pH of 0.005M codeine (C18H21NO3) solution is 9.95. Calculate its ionization constant and pKb. (Given: Antilog 0.95 = 8.913, log1.588 = 0.2)
(ii) Calculate the hydrogen ion concentration in the following biological fluids whose pH are given below:
(a) Human muscle-fluid, 6.83
(b) Human stomach fluid, 1.2
(c) Human blood, 7.38
(Given: antilog 0.17 = 1.48, antilog 0.8 = 6.31, antilog 0.62 = 4.16, antilog 0.6=3.98)
33. (i) Which of the following compounds will not exist as resonance hybrid. Give reason for your answer:
(a) CH3OH
(b) RCONH2
(c) CH3CH2CH=CHNH2
(ii) Explain electrophiles and nucleophiles with the help of one example each. Why does SO3 act as an electrophile?
OR
(i) Which bond is more polar in the following pairs of molecules. Give reason to support your answer.
(a) H3C−H, H3C−Br
(b) H3C−NH2, H3C−OH
(c) H3C−OH, H3C−SH
(ii) Explain the following reaction giving example of each:
(a) Electrophilic addition
(b) Elimination reaction
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
