Class 11 Chemistry
Final Series Sample Paper 5W
Q.1. The stability of +1 oxidation state among Al, Ga, In and Tl increases in the sequence
(a) Al < Ga < In < Tl
(b) Tl < In < Ga < Al
(c) In < Tl < Ga < Al
(d) Ga < In < Al < Tl
Q.2. The basic structural unit of silicates is
(a) SiO32–
(b) SiO42–
(c) SiO–
(d) SiO44–
Q.3. 1 mole of methane (CH4) contains?
(a) 6.022 × 1023 atoms of H
(b) 4 g of hydrogen
(c) 1.81 × 1023 molecules of methane
(d) 4 g of carbon
Q.4. Which of the following statement(s) is not correct?
(a) The electronic configuration of Cr is [Ar] 3d5 4s1.
(b) 'm' can have negative value.
(c) In Ag (47), 23 electrons have one type of spin and 24 electrons have opposite spin.
(d) The oxidation state of nitrogen in HN3 (Hydrazoic acid) is –3.
Q.5. 2.76 g of Ag2CO3 on heating gives a residue weighing
(a) 2.16 g of Ag
(b) 2.32 g of Ag2O
(c) 2.48 g of Ag2O
(d) 2.64 g of Ag2O
Direction (Q.6 to Q.8): In the following questions two or more options may be correct.
Q.6. Which of the following salts are present in hard water?
(a) CaSO4
(b) MgCl2
(c) Ca(HCO3)2
(d) NaHCO3
Or
Which of the following statements about hydrogen is incorrect?
(a) Hydronium ion, H3O+ exists freely in solution.
(b) Dihydrogen does not act as a reducing agent.
(c) Hydrogen has three isotopes of which tritium is the most common.
(d) Hydrogen never acts as a cation in ionic salts.
Q.7. Lead sulphide reacts with hydrogen peroxide to form
(a) PbSO4
(b) H2O
(c) PbSO3
(d) H2
Q.8. Which of the following statements is correct about heavy water?
(a) Its molar mass is 20 g mol–1.
(b) It is obtained by repeated electrolysis of water.
(c) Its density is maximum at 4 °C.
(d) Its density is maximum at 11.4 °C.
Q.9. The metal present in chlorophyll is _________.
Q.10. Electrolysis of molten KCl.MgCl2.6H2O gives _________ and _________.
Q.11. Lithium reacts with nitrogen to form __________.
Q.12. Boron trichloride reacts with lithium aluminium hydride to form diborane, aluminium chloride and lithium chloride. [True/False]
Q.13. Sodium borohydride reduce iodine to form diborane sodium iodide and hydrogen gas. [True/False]
Direction (Q.14 and Q.15): Choose the correct alternative from the following:
(a) Both Assertion and Reason are true and Reason is the correct explanation of Assertion.
(b) Both Assertion and Reason are true but Reason is not the correct explanation of Assertion.
(c) Assertion is true but Reason is false.
(d) Both Assertion and Reason are false.
Q.14. Assertion: SiCl4 reacts with water, CCl4 does not.
Reason: SiCl4 is ionic whereas CCl4 is covalent.
Q.15. Assertion: When CO2 is passed through lime water for long time a white ppt formed gets dissolved.
Reason: Calcium carbonate is insoluble, formed initially, get dissolved due to formation of Ca(HCO3)2.
Q.16. Write the significant figures in the following:
(i) 2.000
(ii) 4.8046
Q.17. Write the electronic configuration of Cu+ (29).
Or
Name the region to which Paschen series belongs?
Q.18. What is bond order of NO?
Q.19. Equal masses of He, O2 and SO2 are taken in closed container. What will be ratio of their partial pressures.
Q.20. For the reaction CO(g) + Cl2(g) ⇌ COCl2(g)
What is the relationship between KP and KC.
Or
Give one example of acidic buffer.
Q.21. 20 mL H2 measured at 15 °C is heated to 250 °C, what is new volume at constant pressure.
Q.22. What is significance of 'a' and 'b' in Vander Waal's equation?
Q.23. State two consequences of phosphate fertilizers causing water pollution.
Q.24. What are pesticides and herbicides? Explain giving examples.
Q.25. How will you convert the following:
(i) Propene to 2,3–Dimethyl butane
(ii) Ethyne to Ethanal.
Q.26. Write IUPAC name of the following compounds:
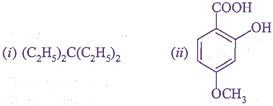
Q.27. (i) Why is second ionisation energy of alkali metals higher than alkaline earth metals?
(ii) What happens when potassium reacts with oxygen? Why?
Or
Complete the following:
(i) CaO + SiO2 →
(ii) Na2CO3 + CO2 + H2O →
Q.28. (a) Define limiting reagent.
(b) If 50 g of CaCO3 is treated with 50 g of HCl, how many grams of CO2 can be produced according to following equation:
CaCO3(s) +2HCl(dil.) → CaCl2(aq) + CO2(g) + H2O(l)
Q.29. The wavelength of first spectral line in the Balmer series is 6561 Å. Calculate the wavelength of the second spectral line in Balmer series.
Or
The radius of first Bohr's orbit of hydrogen atom is 0.529 Å. Calculate the radius of
(i) the third orbit of He+.
(ii) the second orbit of Li2+ ion.
Q.30. (a) Element A, B, C and D have atomic numbers 12, 19, 29 and 36 respectively on the basis of electronic configuration, write which group of the periodic table each element belongs to.
(b) Predict the blocks to which these elements belong. Also predict the periods.
Q.31. (a) Use molecular orbital theory to predict why Be2 molecule does not exist.
(b) Compare the stability of N2+ and N2.
Q.32. Calculate the free energy change at 298 K for the reaction:
Br2(l) + Cl2(g) → 2BrCl(g)
∆H = 29.3 kJ at 298 K. The entropies of Br2(l), Cl2(g) and BrCl(g) at above temperature are 152.3, 223.0, 239.7 J K–1 mol–1 respectively. Predict if the reaction is spontaneous or not.
Q.33. (a) Give an example of decomposition redox reaction.
(b) Balance the following equations:
(i) MnO4– + NO2– + H+ → Mn2+ + NO3– + 4H2O
(ii) Cr2O72– + SO2 + H+ → Cr3+ + SO42– + H2O
Q.34. (i) Why BeSO4 is soluble in water whereas BaSO4 is not?
(ii) Why is lithium considered to be the best reducing agent
(iii) LiI is soluble in ethanol. Why?
Q.35. (a) Equilibrium constant for a reaction is 100. What will be equilibrium constant for reverse reaction?
(b) Write the conjugate acids for Bronsted base Oil and CH3COO–.
(c) Determine the pH of 10–8 M HCl solution considering the H+ produced by water also (log 11 = 1.0414)
Or
(a) What is effect of temperature on value of KSP?
(b) Calculate pH of buffer solution containing 0.01 M solution of NH4OH and 0.1 M solution of NH4Cl. pKb of NH4OH = 4.75.
(c) Classify the following into Acidic and Basic salts.
CH3COONa, NH4Cl, Na2CO3, CuSO4
Q.36. (a) Propanal and Propan–2–one are the ozonolysis products of an alkene. What is the structural formula of the alkene?
(b) Give the major products of the reactions.
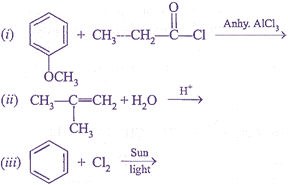
Or
(a) An alkyl halide 'A' of the formula C6H13Cl on treatment with alcoholic KOH give two isomeric alkene (B) and (C) with formula C6H12. Both alkenes on hydrogenation give 2,3–Dimethyl butane. Predict the structures of 'A', 'B' and 'C'.
(b) How is benzene obtained from ethyne? What is process called?
(c) Convert 1–Propanol to 2–Propanol.
Q.37. (a) How is aniline purified and why?
(b) Classify each of the following intermediates

(c) Why is chlorobenzene ortho and para–directing towards electrophilic substitution reaction.
(d) If silver nitrite is added to chlorobenzene, will there be white ppt.
Or
(a) What is function of fusing organic compound with sodium metal?
(b) A student was given the compound C6H4(NH2)SO3H for elemental analysis, while performing Lassaigne's test for nitrogen, which colour will he get and why?
(c) 0.25 g of organic compound containing C, H and O was analysed by combustion method. The increase in mass of calcium chloride tube and potash bulb at the end of operation was found to be 0.15 g and 0.1837 g respectively. Calculate the percentage composition of the compound.
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
