Class 12 Chemistry
Final Series Question Answer Set 1
Q.1. What is the no. of atoms per unit cell (z) in a body-centred cubic structure?
Ans. Contribution
by the atoms present at eight corners = 8 ×  =
1
=
1
Contribution by the atoms present at centre= 1
Total number of atoms present in unit cell= 1 + 1 = 2
Q.2. In reference to surface chemistry, define dialysis.
Ans. Dialysis: The process of removing the dissolved. substances from a colloidal solution by means of diffusion through a suitable membrane is called dialysis.
Q.3. What is the IUPAC name of the complex [Ni(NH3)6]Cl2?
Ans. IUPAC name: Hexaamminenickel (II) chloride.
Q.4. Draw the structure of 3-methylpentanal
Ans.
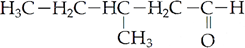
Q.5. Complete the following reaction:
C6H5N2Cl + H3PO2 + H2O →
Ans.
C6H5N2Cl + H3PO2 + H2O → C6H6 + N2 + H3PO3 + HCl
Benzene diazonium Hypophosphorous Benzene Phosphorous
chloride acid acid
Q.6. Define osmotic pressure of a solution. How is the osmotic pressure related to the concentration of a solute in a solution?
Ans. Osmotic pressure: It is the external pressure which is applied on the side solution which is sufficient to prevent the entry of the solvent through semi-permeable membrane.
According to the Boyle-Van't Hoff Law, the osmotic pressure (π) of a dilute solution is directly proportional to its molar concentration provided temperature is constant.
π ∝ C (At constant temperature)
π ∝ CT (At constant concentration)
π = CRT , (R = Solution constant)
or, 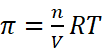
Q.7. Define the following terms:
(i) Half-life of a reaction (t1/2)
(ii) Rate constant (k)
Ans. (i) Half-life of a reaction (t1/2): Half-life period (t1/2) is the time in which half of the substance, has reacted and its concentration is reduced to one-half -of its initial concentration.
(ii) Rate constant (k): Rate constant may be defined as the rate of reaction when the molar concentration of each reactant is taken as unity.
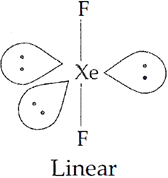
Q.8. Draw the structures of the following:
(i) H2SO4 (ii) XeF2
Ans. (i) Structure of H2SO4
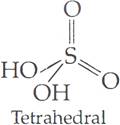
(ii) Structure of XeF2
Q.9. What is meant by disproportionation? Give an example of a disproportionation reaction in aqueous solution.
Ans. Disproportionation: In a disproportionation reaction an element undergoes self-oxidation as well as self-reduction forming two different compounds.
For example, 3MnO42– + 4H+ → 2MnO4– + MnO2 + 2H2O
(VI) (VII) (IV)
Q.10. Suggest reasons for the following features of transition metal chemistry:
(i) The transition metals and their compounds are usually paramagnetic:
(ii) The transition metals exhibit variable oxidation-states.
Ans.
(i) The transition metals and their compounds are usually paramagnetic because of the presence of unpaired electrons in their d-orbitals.
(ii) The transition metals exhibit variable oxidation states because of very close energies of incompletely filled (n – 1) d-orbitals and ns orbitals due to which both can participate in bonding.
Q.11. Explain the mechanism of dehydration steps of ethanol:
CH3CH2OH 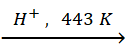 CH2
= CH2 + H2O
CH2
= CH2 + H2O
Ans. Acid Dehydration of ethanol:
CH3CH2OH 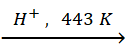 CH2
= CH2 + H2O
CH2
= CH2 + H2O
Mechanism:
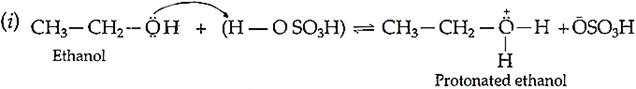
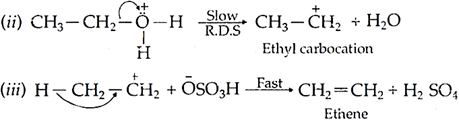
Q.12. Define the following:
(i) Schottky defect
(ii) Frenkel defect
(iii) F-centre
Ans. (i) Schottky defect: If in an ionic crystal of type A+ B–, equal number of cations and anions are missing from their lattice sites so that the electrical neutrality is mainfained, it is called Schottky defect.
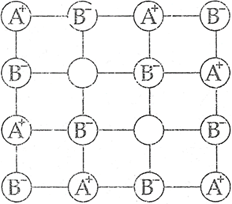
(ii) Frenkel defect: If an ion leaves ifs site from its lattice site and occupies the interstitial site and maintains electrical neutrality, then it is called Frenkel defect.
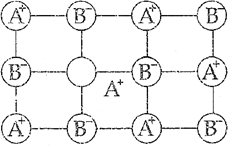
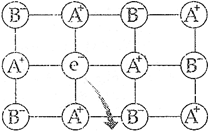
(iii) F-centre The centres which are created by trapping of electrons in anionic vacancies and which are responsible for imparting colour to the crystals are called F-centres.
Q.13. 45 g of ethylene glycol (C2H6O2) is mixed with 600 g of water. Calculate
(i) the freezing point depression
(ii) the freezing point of the solution (Given: Kf of water = l.86 K kg mol–1)
Ans.
(i) Given: w = 45 g, W = 600 g, . Kf = 1.86 K kg mol–1, ∆Tf = ?
Using the formula for freezing point depression,
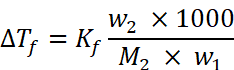
Molar mass of ethylene glycol (C2H6O2), M2 = 2 × 12 + 6 × 1 + 2 × 16 = 62 u
Substituting above values in formula,
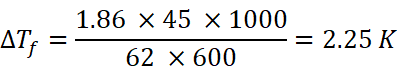
(ii) ∆Tf = Tfo – Tf where, Tfo = freezing point of pure water
=> 2.25 K = 273.15 K – Tf => Tf = 273.15 – 2.28 K
∴ Tf = 270.9 K (Freezing point of the solution)
Q.14. The rate constant of a reaction at 500 K and 700 K are 0.02 s–1 and 0.07 s–1 . Calculate the value of activation energy Ea. (R = 8.314 J K–1 mol–1) respectively.
Ans. Given : k2 = 0.07 s–1, k1 = 0.02 s–1, T1 = 500 K, T2 = 700 K, Ea = ?
Using formula, 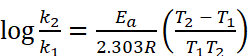
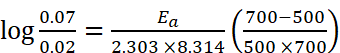
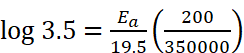
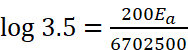
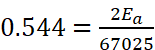
Ea = 18230.8 J
Q.15. Define thefollowing terms:
(i) Electrophoresis
(ii) Adsorption
(iii) Shape selective catalysis
Ans. (i) Electrophoresis: When electric current is passed thtough a colloidal solution, the positiyely charged particles-move towards cathode while negatively charged particles move towards anode where they lose their charge and get coagulated. This phenomenon is known as electrophoresis.
(ii) Adsorption: The phenomenon of accumulation or higher concentration of molecular species (gases or liquids) at the surface rather than in the bulk of a solid or liquid is called adso'rption.
(iii) Shape selective catalysis: The catylytic reaction that depends upon the pore structure of the catalyst and the size of the reactant and product moiecules, is called shapeselective catalysis.
Q.16. Outline the principles of refining of metals by the fcllowing methods:
(i) Distillation
(ii) Zone refining
(iii) Electrolysis
Ans. (i) Principle of Distillation: The impure metal is evaporated to obtain the pure metal as distillate. The metals having low boiling points (Bi, Hg, Cd etc.) readily change into vapours leaving behind impurities which further get collected in receivers and upon cooling, pure metal is obtained.
(ii) Zone refining of metals: It is based on the principle that the impurities are more soluble in melt than in the solid state of the metal.
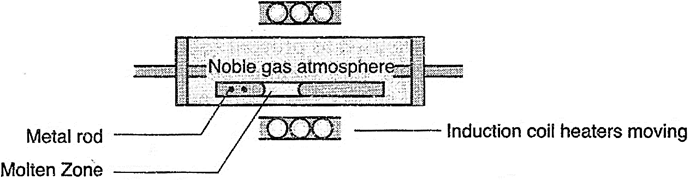
(iii) Electrolysis: Here the impure metal is J!lade to act as 9node and a strip of the same metal in pure form is used as cathode. When they both are put in a suitable electrolyte containing soluble salt of same metal, the more basic metal remains in the solution and the less basic ones go to the anode mud.
Example : In refining of Cu
At anode : (oxidation)
Cu → Cu2+ + 2e–
At cathode : (reduction)
Cu2+ + 2e– → Cu
Q.17. Write down the reactions taking place in different zones in the blast furnace during the extraction of iron. How is pig iron different from cast iron?
Ans.
Reactions occur in blast furnace in the extraction of iron from iron oxide ores. The following oxidations occur in different zones of furnace:
(i) Zone of combustion
C + O2 → CO2 + Heat (Exothermic)
(ii) Zone of heat absorption (lower part)
CO2 + C → 2CO + Heat (Endothermic)
(iii) Zone of slag formation (middle part of furnace)
CaCO3  CaO
+ CO2
CaO
+ CO2
CaO + SiO2 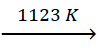 CaSiO3
CaSiO3
(iv) Zone of reduction (upper part) ores are reduced to FeO and CuO
Fe2O3 +
CO 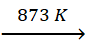 2FeO
+ CO2
2FeO
+ CO2
3Fe2O3 +
CO 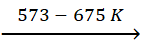 2Fe3O4
+ CO2
2Fe3O4
+ CO2
Fe3O4 +
CO 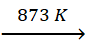 3FeO
+ CO2
3FeO
+ CO2
FeO + CO  Fe
+ CO2
Fe
+ CO2
Fe2O3 +
3C  2Fe
+ 3CO
2Fe
+ 3CO
Pig iron differs from cast iron with respect to the carbon contents. Pig iron has nearly 4% carbon content and cast iron has nearly 3% carbon content. Cast iron is hard and brittle whereas pig iron is soft.
Q.18. What is lanthanoid contraction? What are the consequences of lanthanoid contraction?
Ans. Lanthanoid contraction: The overall decrease in atomic and ionic radii with increasing atomic number from La to Lu due to imperfect shielding of 4f-orbital is known as lanthanoid contraction.
Cause: As we move along the lanthanoid series, the effective nuclear charge increases on addition of electrons and the electrons added in f-subshell causes imperfect shielding which is unable to counterbalance the effect of the increased nuclear charge. Hence the contraction in size occurs.
Consequences:
(i) Due to small change in atomic radii, the chemical properties of lanthanoids are very similar due to which separation of lanthanoids becomes very difficult.
(ii) There is similarity in size of e1ements belonging to same group of second and third transition series.
Example: Zr and Hf are known as chemical twins due to their almost identical radii.
Q.19. Indicate the types of isomerism exhibited by the following complexes.
(i) (Co(NH3)5(NO2)]2+
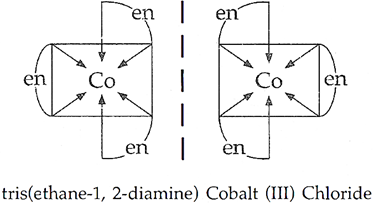
(ii) [Co(en)3]Cl3 (en = ethylene diamine)
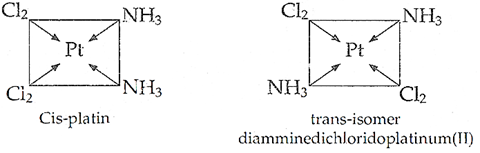
(iii) [Pt(NH3)2Cl2]
Ans. (i) Linkage isomerism is shown by [Co(NH3)5(NO2)]2+
[Co(NH3)5(NO2)]2+ [Co(NH3)5(ONO)]2+
Pentaamminenitro Cobalt (III) Pentaamminenitrito-N-Cobalf(III)
(ii) [Co(en)3]Cl3 shows optical isomerism
(iii) [Pt(NH3)2Cl2] shows geometrical isomerism.
Q.20. Name the following according to IUPAC system:
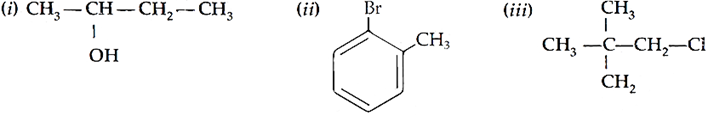
Ans. (i) Butan-2-ol
(ii) 2-Bromotoluene
(iii) 1-Chloro-2,2-dimethylpropane (2,2-dimethylchloropropane)
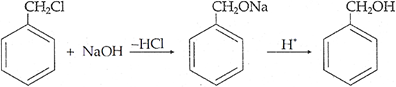
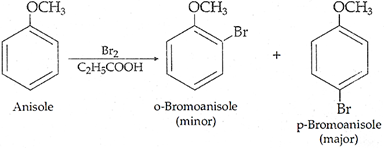
Q.21. How are the fol1owing conversions carried out?
(i) Propene to propane-2-ol
(ii) Benzyl chloride to Benzyl alcohol
(iii) Anisole to p-Bromoanisole
Ans.
(i)
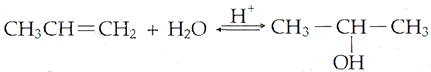
(ii)
(iii)
Q.22. An aromatic compound ‘A’ on treatment with aqueous ammonia and heating forms compound ‘B’ ( which on heating with Br2 and KOH forms a compound ‘C’ of molecular formula C6H7N. Write the structures and IUPAC names of compounds A, B and C.
Ans. The data shows that C6H7N may be C6H5NH2 i.e. Aniline. Since it is obtained by heating with Br2 and KOH (Hoffmann bromamide reaction), then the compound 'B' is Benzamide C6H5CONH2 which is in turn obtained by reaction with aqueous -ammonia then the compound 'A' can be Benzoic acid i.e. C6H5COOH.
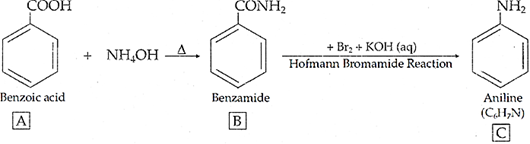
Thus,
A → Benzoic acid, B → Benzamide, C → Aniline
Q.23. How are vitamins classified? Name the vitamin responsible for the coagulation of blood.
Ans. Vitamins are classified into two types:
(i) Water insoluble vitamins: These are fat soluble substances E.g. Vitamin A, D, E and K.
(ii) Water soluble vitamins: These include Vitamin B-Complex (except B12) and Vitamin C. Vitamin K or phylloquinone is responsible for the coagulation of blood.
Q.24. Write the names and structures of the monomers of the following polymers.
(i) Buna-S
(ii) Neoprene
(iii) Teflon
Ans.
(i) Buna-S: 1, 3-butadiene and Styrene
(ii) Neoprene: Chloroprene
(iii) Teflon: tetrafluoroethene (TFE)
Q.25. Sam went to a departmental store to purchase groceries. On one of the shelves he noticed sugar-free tablets. He decided to buy them for his grandfather who was a diabetic. There were three types of sugar-free tablets. Sam decided to buy sucrolose which was good for his grandfather's health.
(i) Name another sugar free tablet which Sam did not buy.
(ii) Was it right to purchase such medicines without doctor’s prescription?
(iii) What quality of Sam is reflected above?
Ans. (i) Other tablets are Aspartame and Saccharin.
(ii) No, it was not right because medicines should always be taken under medical supervision as a doctor knows best about your body reactivity towards medicines.
(iii) Sam was aware of the damage caused by diabeties and had good knowledge of the benefits of artificial sweeteners. He was concerned about his grandfather's health and empathised with him.
Q.26. (a) Define the following terms:
(i) Molar conductivity (Λm)
(ii) Secondary batteries
(iii) Fuel cell
(b) State the following laws:
(i) Faraday first law of electrolysis
(ii) Kohlrausch's law of independent migration of ions
Ans.
(a) (i) Molar conductivity (Λm) :Molar conductivity can be defined as the conductance of the volume V of electrolytic solution kept between two electrodes of a conducting cell at distance of uriit length but. havmg area of cross section large enough to accomodate sufficient volume of solution that contains one mole of the electrolyte.
Λm = KV
(ii) Secondary batteries: Those cells which can be recharged on passing electric current through them in opposite direction and can be used again are called secondary batteries. e.g. Lead-acid storage cell.
(iii) Fuel cell : Galvanic cells that are designed to convert the chemical energy of combustion of fuels like hydrogen, methane etc. into electrical energy are called fuel cells. e.g. H2 – O2 fuel cell.
(i) Faraday first law of electrolysis: According to this law the mass of the substance deposited or liberated at any electrode during electroly'Sis is directly proportional to the quantity of charge passed through the electrolyte.
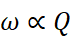
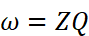
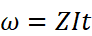
(ii) Kohlrausch's law of independent migration of ions: According to this law limiting-molar conductivity of an electrolyte can be represcnted as the sum of the limiting ionic conductivities of the cation and the anion each multiplied) with the number of ions present in one formula unit of electrolyte.
Λmo for AxBy = xl+o + yl–o
Q.27. (a) Define the term degree of dissociation. Write an expression that relates, the molar conductivity of a weak electrolyte to its degree of dissociation.
(b) For the cell reaction
Ni(s) | Ni2+(aq) || Ag+(aq) | Ag(s)
Calculate the equilibrium constant at 25 °C. How much maximum work would be obtained by operation of this cell?
EoNi2+/Ni = 0.25 V and EoAg+/Ag = 0.80 V
Ans.
(a) Degree of dissociation: It is the measure of the extent to which an electrolyte gets dissociated into its constitutent ions.
Thus higher the degree of dissociation higher will be its molar conductance.
Mathematically it can be expressed as:
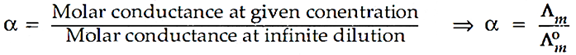
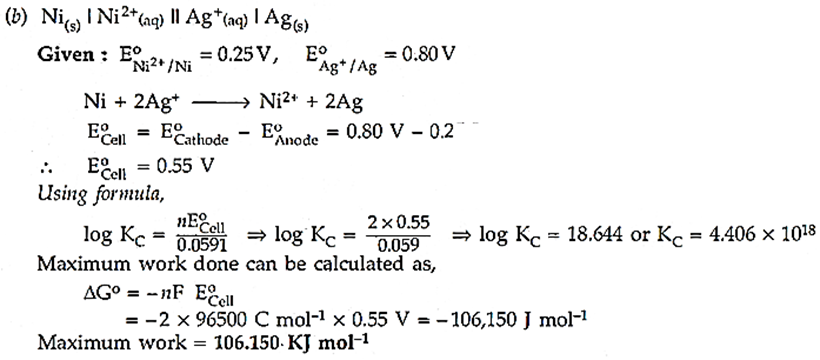
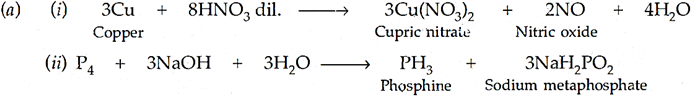
Q.28. (a) Complete the following chemical equations:
(i) Cu + HNO3 (dilute) →
(ii) P4 + NaOH + H2O →
(b) (i) Why does R3P = O exist but R3N = O does not? (R = alkyl group)
(ii) Why is dioxygen a gas but sulphur a solid?
(iii) Why are halogens coloured?
Ans.
(b) (i) Nitrogen in R3N = O cannot form pp - dp multiple bonds because it cannot expand tts covalency beyond 14 due to absence of d-orbitalr so it will not exist. But R3P = O can do so due to presence of d-orbitals and formation of pp - dp multiple bonds which can expand its covalency up to 5.
(ii) Because of bigger size and the strong forces of attraction holding 8 atoms, their bonds cannot be broken easily and hence sulphur exist's as solid, while oxygen due to high electro-negativity and tendency to form pp - dp multiple bonds through Vander-waals forces of attraction can be broken easily ahd hePce exists as gas.
(iii) All halogens are coloured due to absorption of light in the visible region as a result of which their electrons get excited to higher energy levels and while returning to lower level transmit energy of cotresponding colour.
Q.29. (a) Write balanced equations for the following reactions:
(i) Chlorine reacts with dry slaked lime.
(ii) Carbon reacts with concentrated H2SO4.
(b) Describe the contact process for the manufacture of sulphuric acid with special reference to the reaction conditions, catalysts used and the yield in the process.
Ans.
(a) (i) Chlorine reacts with dry slaked lime
2Ca(OH)2 + 2Cl2 → Ca(OCl)2 + CaCl2 + 2H2O
Calcium hydroxide Calcium oxychloride
(ii) Carbon reacts with concentrated H2SO4
C + 2H2SO4 (conc.) → CO2 + 2SO2 + 2H2O
(b) Contact process of sulphuric acid:
It involves the following steps:
(i) Formation of sulphur dioxide by burning either sulphur or iron pyrites in excess of air.
S + O2 → SO2
4FeS2 + 11O2 → 2Fe2O3 + 8SO2
(ii) Catalytic oxidation of SO2 into SO3 by using V2O5 as catalyst
2SO2 (g) + O2 (g) 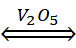 SO3(g)
∆H = –196.6 KJ mol–1
SO3(g)
∆H = –196.6 KJ mol–1
According to Le-Chatelier' s principle the reaction conditions are :
(a) High concentration of reactants
(b) Low temperature (623-723 K)
(c) High pressure (2 bar)
By obeying above conditions the yield of H2SO4 will be 96-98%.
(iii) Absorption of SO3 in 98% H2SO4 to give Oleum (H2S2O7)
SO3 + H2SO4 → H2S2O7
(iv) Dilution of Oleum to give sulphuric acid
H2S2O7 + H2O → 2H2SO4
Q.30. (a) Describe the following giving chemical equations:
(i) De-carboxylation reaction (ii) Friedel-Crafts reaction.
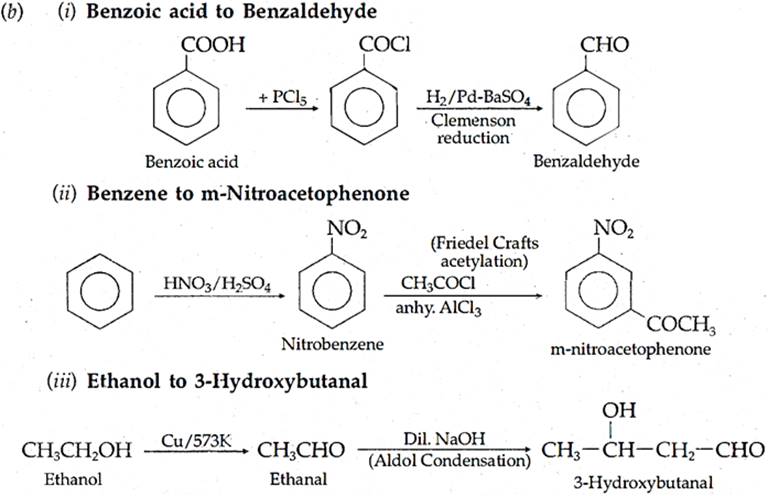
(b) How will you bring about the following conversions?
(i) Benzoic acid to Benzaldehyde (ii) Benzene to m-Nilroacetophenone
(iii) Ethanol to 3-Hydroxybutanal
Ans. (a) (i) De-carboxylation reaction: When sodium salt of carboxylic acid is heated with soda lime (NaOH + CaO), then corresponding alkane and CO2 will be evolved.
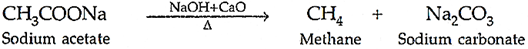
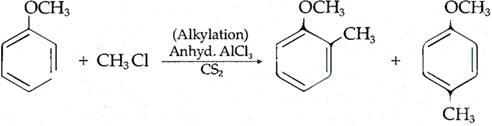
(ii) Friedel-Crafts reaction: The introduction of alkyl or acetyl group in the presence of anhydrous almuninium chloride (AlCl3) as catalyst to ortho and para positions of an aromatic compound is called Friedel-Crafts reaction.
Online Tuitions & Self-Study Courses for Grade 6 to 12 & JEE / NEET
