Q.1. An aqueous solution of compound A gives ethane on electrolysis. The compound A is
(a ) Ethyl acetate
(b) Sodium acetate
(c) Sodium propionate
(d) Sodium ethoxide.
Q.2. Arrange the following in decreasing order of their boiling points.
(A) n−butane (B) 2−methylbutane
(C) n−pentane (D) 2, 2−dimethylpropane
(a) A > B > C > D
(b) B > C > D > A
(c) C > B > D > A
(d) D > C > B > A
Q.3. Isopropyl bromide on Wurtz reaction gives
(a) Hexane
(b) Propane
(c) 2, 3−Dimethylbutane
(d) Neohexane
Q.4. The correct IUPAC name of the following alkane is
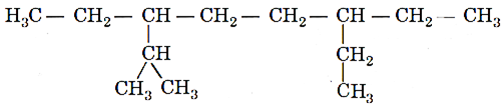
(a) 3, 6−Diethyl−2−methyloctane
(b) 3−Isopropyl−6−ethyloctane
(c) 3−Ethyl−5−isopropyloctane
(d) 5−Isopropyl−3−ethyloctane
Q.5. Which of the following has least boiling point ?
(a) n−Hexane
(b) n−Pentane
(c) 2−Methylbutane
(d) 2,2−Dimethyl propane
Q.6. The chlorination of methane is an example of
(a) elimination reaction
(b) substitution reaction
(c) addition reaction
(d) oxidation reaction.
Q.7. Arrange the halogens F2, Cl2, Br2, I 2, in order of their increasing reactivity with alkanes.
(a) I2 < Br2 < Cl2 < F2
(b) Br2 < I2 < Cl2 < F2
(c) Br2 < Cl2 < F2 < I2
(d) F2 < Cl2 < Br2 < I2
Q.8. In the reaction,
![]()
(a) CH3CHO
(b) CH4
(c) C2H6
(d) CH3COCH3
Q.9. Which of the following can exhibit geometrical isomerism ?
(a) But−2−yne
(b) But−2−ene
(c) But−1−ene
(d) Butan−2−ol
Q.10. The compound which forms only acetaldehyde upon ozonolysis is
(a) Ethene
(b) Propyne
(c) But−1−ene
(d) But−2−ene
Q.11. In the reaction:
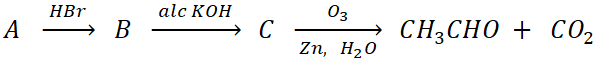
the compound A is
(a) Ethylene
(b) Acetic acid
(c) Propene
(d) 1−Butene
Q.12. Which of the following will not show geometrical isomerism?
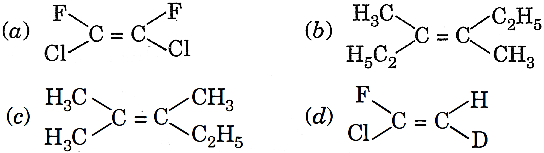
Q.13. The ozonolysis of (CH3)2C=C(CH3)2 followed by treatment with zinc and water will give
(a) acetone
(b) acetaldehyde and acetone
(c) acetic acid
(d) formaldehyde
Q.14. Ethylene reacts with Baeyer's reagent to give
(a) glycol
(b) acetaldehyde
(c) oxalic acid
(d) ethyl alcohol
Q.15. A gas on passing through ammoniacal AgNO3 solution does not give any precipitate but decolourises alkaline KMnO4 solution. The gas may be
(a) C2H6
(b) C2H4
(c) C2H2
(d) C3H8
Q.16. When 3, 3−dimethyl−2−butanol is heated with H 2SO4, the major product is
(a ) 3, 3−dimethylbut−1−ene
(b) 2, 3−dimethylbut−2−ene
(c) 2, 3−dimethylbut−1−ene
(d) cis and trans isomers of 2, 3−dimethylbut−2−ene
Q.17. Maleic acid and fumaric acid are the forms of
(a) Chain isomers
(b) Conformations
(c) Geometrical isomers
(d) Optical isomers
Q.18. Which of the following has smallest heat of hydrogenation per mole ?
(a) But−1−ene
(b) Trans− But−2−ene
(c) Cis− But−2−ene
(d) Buta−1,3−diene
Q.19. Reaction of HBr with propene in the presence of peroxide gives
(a) 3−Bromopropane
(b) alkyl bromide
(c) n−propyl bromide
(d) isopropyl bromide
Q.20. Oxidation of an alkene X gives a diol; further oxidation gives a diketone. Which one of the following could be 'X' ?
(a) (CH3)2C=C(CH3)2
(b) CH3CH=C(CH3)2
(c) (CH3)2CHCH=CH2
(d) C6H5CH=CHC6H5
Q.21. Ethyne adds a molecule of methyl alcohol in the presence of alkali to give
(a) Acetone
(b) Methyl vinyl ether
(c) Acetaldehyde
(d) Acetic acid
Q.22. Which reagent can be used to distinguish between but−1−yne and but−2−yne ?
(a) alc. KOH
(b) alc. KMnO4
(c) Br2 water
(d) Ag+
Q.23. Acetylene gas is obtained by the electrolysis of
(a) sodium fumarate
(b) sodium succinate
(c) sodium maleate
(d) both (a) and (c)
Q.24. Arrange the following carbanions in order of their decreasing stability.
(A) H3C − C ≡ C − (B) H − C ≡ C − (C) H3C – CH2−
(a) B > A > C
(b) A > B > C
(c) C > A > B
(d) C > B > A
Q.25. Which of the following will not react with ammoniacal solution of silver nitrate ?
(a) CH3C ≡ CH
(b) (CH3)2CH−C ≡ CH
(c) CH3C ≡ CCH 3
(d) HC ≡ CH
Q.26. In the reaction
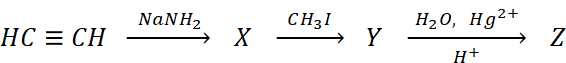
Z is :
(a) CH3CHO
(b) CH3CH2CH=CH2
(c) CH3COCH3
(d) CH3CH2CHO
Q.27. Number of acidic hydrogen atoms in but−1−yne is
(a) 1
(b) 2
(c) 3
(d) 4
Q.28. The alkyne which will react with KMnO4 to give pyruvic acid is
(a) Ethyne
(b) Propyne
(c) Butyne
(d) Pent−2−yne
Q.29.
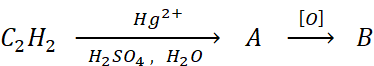
The compound B is
(a) an acid
(b) an aldehyde
(c) ketone
(d) ethanol
Q.30. Products of the reactions are
![]()
(a) CH3COOH + CO2
(b) CH3COOH + HOOCCH2CH3
(c) CH3CHO + CH3CH2CHO
(d) CH3COOH + CH3COCH3
Q.31. In benzene, each carbon atom undergoes
(a) sp
(b) sp2
(c) sp3
(d) sp2 and sp3 hybridisation
Q.32. Benzene reacts with C6H5COCl in the presence of anhydrous AlCl3 to give
(a) toluene
(b) naphthalene
(c) acetophenone
(d) benzophenone
Q.33. The number of π−bonds in naphthalene is
(a) 5
(b) 6
(c) 3
(d) 4
Q.34. Benzene molecule has
(a) 6 a and 6 1t bonds
(b) 16 a and 6 1t bonds
(c) 12 a and 3 1t bonds
(d) 6 a and 3 1t bonds.
Q.35. Benzene reacts with acetyl chloride in the presence of AlCl3 to give
(a) acetophenone
(b) toluene
(c) benzophenone
(d) ethyl benzene.
Q.36. An aromatic compound X, C7H7Cl on oxidation gives an aromatic compound Y. The soda−lime decarboxylation of Y gives benzene. X is
(a) o−chlorotoluene
(b) p−chlorotoluene
(c) benzyl chloride
(d) m−chlorotoluene
Q.37. Which of the following is less reactive than benzene towards electrophilic substitution reactions ?
(a) Nitrobenzene
(b) Aniline
(c) Bromo benzene
(d) Chlorobenzene
Q.38. In the reaction,
C6H6 + RCOCl ![]() C6H5COR + HCl
C6H5COR + HCl
the attacking electrophilic is
(a) AlCl2+
(b) R+
(c) RCO+
(d) AlCl4
Q.39. n−Propyl chloride and benzene react in the presence of anhydrous AlCl 3 to form.
(a) ethyl benzene
(b) methyl benzene
(c) n−propyl benzene
(d) iso− propyl benzene
Q.40. The attacking reagent in electrophilic sulphonation of benzene is
(a) SO42−
(b) SO32−
(c) SO2
(d) SO3
Q.41. Benzene can be converted into acetophenone by treating it with
(a) Acetone in the presence of HCl
(b) Acetyl chloride in the presence of AlCl3
(c) Methyl chloride in the presence of AlCl3
(d) Acetaldehyde in the presence of Fe
Q.42. Which of the following is not o− and p−directing group ?
(a) −Cl
(b) −NH2
(c) −CH3
(d) −NO2
Q.43. Which of the following does not obey Huckel rule ?
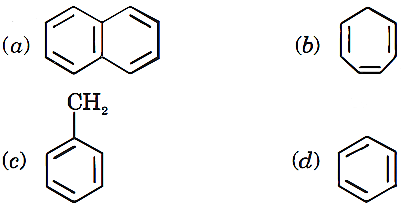
Q.44. In Friedel Craft reaction, anhydrous AlCl3 is used. Its function is to
(a) absorb HCl
(b) absorb H2O
(c) produce electrophile
(d) produce nucleophile
Q.45. The electrophile in nitration of benzene reaction is
(a) NO2
(b) NO2−
(c) NO2+
(d) NO
Answers
A.1: (b)
A.2: (c)
A.3: (c)
A.4: (a)
A.5: (d)
A.6: (b)
A.7: (a)
A.8: (b)
A.9: (b)
A.10: (d)
A.11: (c)
A.12: (c)
A.13: (a)
A.14: (a)
A.15: (b)
A.16: (b)
A.17: (c)
A.18: (b)
A.19: (c)
A.20: (d)
A.21: (b)
A.22: (d)
A.23: (d)
A.24: (a)
A.25: (c)
A.26: (c)
A.27: (a)
A.28: (b)
A.29: (a)
A.30: (b)
A.31: (b)
A.32: (d)
A.33: (a)
A.34: (c)
A.35: (a)
A.36: (c)
A.37: (a)
A.38: (c)
A.39: (d)
A.40: (d)
A.41: (b)
A.42: (d)
A.43: (b)
A.44: (c)
A.45: (c)
Online Tuitions and Self-Study Courses for Grade 6 to 12 & JEE / NEET
