Q.1. Explain why Grignard's reagents should be prepared under anhydrous conditions.
Q.2. Predict the order of reactivity of four isomeric bromobutanes in SN1 reaction.
Q.3. Which would undergo SN2 reaction faster in the following pair and why?
CH3-CH2-Br and CH3-CH2-I
Q.4. Write the structure of an isomer of compound C4H9Br which is most reactive towards SN1 reaction.
Q.5. Out of chlorobenzene and benzyl chloride, which one gets easily hydrolysed by aqueous NaOH and why?
Q.6. Out of 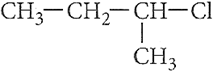 and
and 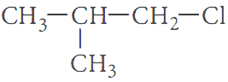 which is more
reactive towards SN1 reaction and why?
which is more
reactive towards SN1 reaction and why?
Q.7. Which would undergo SN2 reaction faster in the following pair and why?
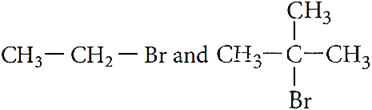
Q.8. Which would undergo SN1 reaction faster in the following pair and why?
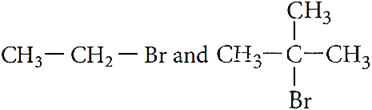
Q.9. Identify the chiral molecule in the following pair:
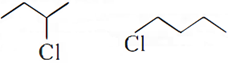
Q.10. Which halogen compound in each of the following pairs will react faster in SN2 reaction:
(i) CH3Br or CH3I
(ii) (CH3)3CCl or CH3Cl
Q.11. What happens when CH3-Br is treated with KCN?
Q.12. What happens when ethyl chloride is treated with aqueous KOH?
Q.13. Why is (±)-butan-2-ol is optically inactive?
Q.14. Which compound in the following pair undergoes faster SN1 reaction?
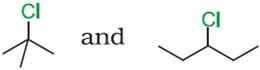
Q.15. How may methyl bromide be preferentially converted to methyl isocyanide?
Online Tuitions and Self-Study Courses for Grade 6 to 12 & JEE / NEET
